Dendritic cell immunizations alone or combined with low doses of interleukin-2 induce specific immune responses in melanoma patients
- PMID: 16297169
- PMCID: PMC1809550
- DOI: 10.1111/j.1365-2249.2005.02948.x
Dendritic cell immunizations alone or combined with low doses of interleukin-2 induce specific immune responses in melanoma patients
Abstract
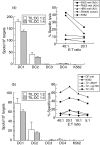
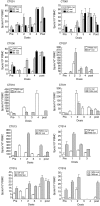
Dendritic cell (DC)-based therapy has proved to be effective in patients with a variety of malignancies. However, an optimal immunization protocol using DCs and the best means for delivering antigens has not yet been described. In this study, 20 patients with malignant melanoma in stages III or IV were vaccinated with autologous DCs pulsed with a melanoma cell lysate, alone (n = 13) or in combination with low doses of subcutaneous (s.c.) interleukin (IL)-2 injections (n = 7), to assess toxicity, immunological and clinical responses. Monocyte-derived DCs were morphological, phenotypic and functionally characterized in vitro. Peripheral blood mononuclear cells (PBMC), harvested from patients either prior to and after the treatment, were analysed using enzyme-linked immunosorbent spot (ELISPOT). After vaccination, 50% of the patients tested (seven of 13) from the first group and (three of seven) from the second, showed an increase in interferon (IFN)-gamma production in response to allogeneic melanoma cell lines but not to controls. Four of five tested human leucocyte antigen (HLA)-A2(+) patients with anti-melanoma activity also showed specific T cell responses against peptides derived from melanoma-associated antigens. Delayed type IV hypersensitivity reaction (DTH) against melanoma cell lysate was observed in six of 13 patients from the group treated with DC vaccines only and four of seven from the group treated with the combination of DCs and IL-2. Significant correlations were found between DTH-positive responses against tumour lysate and both disease stability and post-vaccination survival on the stage IV patients. There were no toxicities associated with the vaccines or evidence of autoimmunity including vitiligo. Furthermore, no significant enhancement was observed as a result of combining DC vaccination with IL-2. Our data suggest that autologous DCs pulsed with tumour lysate may provide a standardized and widely applicable source of melanoma specific antigens for clinical use. It is safe and causes no significant side effects and has been demonstrated to be partially efficient at triggering effective anti-melanoma immunity.
Figures



Similar articles
-
Dendritic cells loaded with killed allogeneic melanoma cells can induce objective clinical responses and MART-1 specific CD8+ T-cell immunity.J Immunother. 2006 Sep-Oct;29(5):545-57. doi: 10.1097/01.cji.0000211309.90621.8b. J Immunother. 2006. PMID: 16971810 Clinical Trial.
-
Maturation of dendritic cells is a prerequisite for inducing immune responses in advanced melanoma patients.Clin Cancer Res. 2003 Nov 1;9(14):5091-100. Clin Cancer Res. 2003. PMID: 14613986
-
Immunohistological analysis of peptide-induced delayed-type hypersensitivity in advanced melanoma patients treated with melanoma antigen-pulsed mature monocyte-derived dendritic cell vaccination.J Dermatol Sci. 2009 Jan;53(1):40-7. doi: 10.1016/j.jdermsci.2008.07.013. Epub 2008 Sep 19. J Dermatol Sci. 2009. PMID: 18804963 Clinical Trial.
-
[Advances in cellular immunotherapy for malignant melanoma].Rev Med Chil. 2004 Sep;132(9):1115-26. Rev Med Chil. 2004. PMID: 15543770 Review. Spanish.
-
TAPCells, the Chilean dendritic cell vaccine against melanoma and prostate cancer.Biol Res. 2013;46(4):431-40. doi: 10.4067/S0716-97602013000400014. Biol Res. 2013. PMID: 24510145 Review.
Cited by
-
Adjuvant Autologous Melanoma Vaccine for Macroscopic Stage III Disease: Survival, Biomarkers, and Improved Response to CTLA-4 Blockade.J Immunol Res. 2016;2016:8121985. doi: 10.1155/2016/8121985. Epub 2016 May 18. J Immunol Res. 2016. PMID: 27294163 Free PMC article. Clinical Trial.
-
Tumor lysate-based vaccines: on the road to immunotherapy for gallbladder cancer.Cancer Immunol Immunother. 2018 Dec;67(12):1897-1910. doi: 10.1007/s00262-018-2157-5. Epub 2018 Mar 29. Cancer Immunol Immunother. 2018. PMID: 29600445 Free PMC article. Review.
-
Lithraea caustic (Litre) Extract Promotes an Antitumor Response Against B16 Melanoma.Front Pharmacol. 2019 Oct 22;10:1201. doi: 10.3389/fphar.2019.01201. eCollection 2019. Front Pharmacol. 2019. PMID: 31695610 Free PMC article.
-
Haptoglobin Induces a Specific Proteomic Profile and a Mature-Associated Phenotype on Primary Human Monocyte-Derived Dendritic Cells.Int J Mol Sci. 2022 Jun 21;23(13):6882. doi: 10.3390/ijms23136882. Int J Mol Sci. 2022. PMID: 35805888 Free PMC article.
-
Long-Term Survival and Immune Response Dynamics in Melanoma Patients Undergoing TAPCells-Based Vaccination Therapy.Vaccines (Basel). 2024 Mar 27;12(4):357. doi: 10.3390/vaccines12040357. Vaccines (Basel). 2024. PMID: 38675738 Free PMC article.
References
-
- Harris M. Monoclonal antibodies as therapeutic agents for cancer. Lancet Oncol. 2004;5:292–302. [Review] - PubMed
-
- Rosenberg SA, Lotze MT, Yang JC, et al. Combination therapy with interleukin-2 and alpha-interferon for the treatment of patients with advanced cancer. J Clin Oncol. 1989;7:1863–74. - PubMed
-
- Lotze MT, Matory YL, Rayner AA, et al. Clinical effects and toxicity of interleukin-2 in patients with cancer. Cancer. 1986;58:2764–72. - PubMed
-
- Rosenberg SA, Lotze MT, Muul LM, et al. Observations on the systemic administration of autologous lymphokine-activated killer cells and recombinant interleukin-2 to patients with metastatic cancer. N Engl J Med. 1985;313:1485–92. - PubMed
-
- Rosenberg SA, Spiess P, Lafreniere R. A new approach to the adoptive immunotherapy of cancer with tumour-infiltrating lymphocytes. Science. 1986;233:1318–22. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

