Modulation of HLA-G expression in human neural cells after neurotropic viral infections
- PMID: 16306594
- PMCID: PMC1316015
- DOI: 10.1128/JVI.79.24.15226-15237.2005
Modulation of HLA-G expression in human neural cells after neurotropic viral infections
Abstract
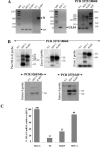
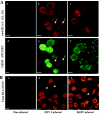
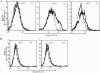
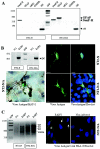
HLA-G is a nonclassical human major histocompatibility complex class I molecule. It may promote tolerance, leading to acceptance of the semiallogeneic fetus and tumor immune escape. We show here that two viruses-herpes simplex virus type 1 (HSV-1), a neuronotropic virus inducing acute infection and neuron latency; and rabies virus (RABV), a neuronotropic virus triggering acute neuron infection-upregulate the neuronal expression of several HLA-G isoforms, including HLA-G1 and HLA-G5, the two main biologically active isoforms. RABV induces mostly HLA-G1, and HSV-1 induces mostly HLA-G3 and HLA-G5. HLA-G expression is upregulated in infected cells and neighboring uninfected cells. Soluble mediators, such as beta interferon (IFN-beta) and IFN-gamma, upregulate HLA-G expression in uninfected cells. The membrane-bound HLA-G1 isoform was detected on the surface of cultured RABV-infected neurons but not on the surface of HSV-1-infected cells. Thus, neuronotropic viruses that escape the host immune response totally (RABV) or partially (HSV-1) regulate HLA-G expression on human neuronal cells differentially. HLA-G may therefore be involved in the escape of certain viruses from the immune response in the nervous system.
Figures

Similar articles
-
Modulation of HLA-G and HLA-E expression in human neuronal cells after rabies virus or herpes virus simplex type 1 infections.Hum Immunol. 2007 Apr;68(4):294-302. doi: 10.1016/j.humimm.2006.12.003. Epub 2006 Dec 28. Hum Immunol. 2007. PMID: 17400066
-
A functional role of HLA-G expression in human gliomas: an alternative strategy of immune escape.J Immunol. 2002 May 1;168(9):4772-80. doi: 10.4049/jimmunol.168.9.4772. J Immunol. 2002. PMID: 11971028
-
Human leukocyte antigen G up-regulation in lung cancer associates with high-grade histology, human leukocyte antigen class I loss and interleukin-10 production.Am J Pathol. 2001 Sep;159(3):817-24. doi: 10.1016/S0002-9440(10)61756-7. Am J Pathol. 2001. PMID: 11549573 Free PMC article.
-
HLA-G protein processing and transport to the cell surface.Cell Mol Life Sci. 2002 Sep;59(9):1460-6. doi: 10.1007/s00018-002-8521-8. Cell Mol Life Sci. 2002. PMID: 12440768 Free PMC article. Review.
-
Does 'soluble' HLA-G really exist? Another twist to the tale.Mol Hum Reprod. 2005 Oct;11(10):695-8. doi: 10.1093/molehr/gah196. Epub 2005 Dec 5. Mol Hum Reprod. 2005. PMID: 16330473 Review.
Cited by
-
Emerging topics and new perspectives on HLA-G.Cell Mol Life Sci. 2011 Feb;68(3):433-51. doi: 10.1007/s00018-010-0584-3. Epub 2010 Nov 16. Cell Mol Life Sci. 2011. PMID: 21080027 Free PMC article. Review.
-
Role of non-classical MHC class I molecules in cancer immunosuppression.Oncoimmunology. 2013 Nov 1;2(11):e26491. doi: 10.4161/onci.26491. Epub 2013 Oct 21. Oncoimmunology. 2013. PMID: 24482746 Free PMC article. Review.
-
Neurons under viral attack: victims or warriors?Neurochem Int. 2010 May-Jun;56(6-7):727-35. doi: 10.1016/j.neuint.2010.02.016. Epub 2010 Mar 4. Neurochem Int. 2010. PMID: 20206655 Free PMC article. Review.
-
Ambivalent role of the innate immune response in rabies virus pathogenesis.J Virol. 2011 Jul;85(13):6657-68. doi: 10.1128/JVI.00302-11. Epub 2011 Apr 27. J Virol. 2011. PMID: 21525357 Free PMC article.
-
IL-10 reduces levels of apoptosis in Toxoplasma gondii-infected trophoblasts.PLoS One. 2013;8(2):e56455. doi: 10.1371/journal.pone.0056455. Epub 2013 Feb 13. PLoS One. 2013. PMID: 23418570 Free PMC article.
References
-
- Anglen, C. S., M. E. Truckenmiller, T. D. Schell, and R. H. Bonneau. 2003. The dual role of CD8+ T lymphocytes in the development of stress-induced herpes simplex encephalitis. J. Neuroimmunol. 140:13-27. - PubMed
-
- Baloul, L., S. Camelo, and M. Lafon. 2004. Up-regulation of Fas ligand (FasL) in the central nervous system: a mechanism of immune evasion by rabies virus. J. Neurovirol. 10:372-382. - PubMed
-
- Becher, B., A. Prat, and J. P. Antel. 2000. Brain-immune connection: immuno-regulatory properties of CNS-resident cells. Glia 29:293-304. - PubMed
-
- Cabello, A., A. Rivero, M. J. Garcia, J. M. Lozano, J. Torre-Cisneros, R. Gonzalez, G. Duenas, M. D. Galiani, A. Camacho, M. Santamaria, R. Solana, C. Montero, J. M. Kindelan, and J. Pena. 2003. HAART induces the expression of HLA-G on peripheral monocytes in HIV-1 infected individuals. Hum. Immunol. 64:1045-1049. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

