Overexpression of tumor necrosis factor alpha by a recombinant rabies virus attenuates replication in neurons and prevents lethal infection in mice
- PMID: 16306612
- PMCID: PMC1316002
- DOI: 10.1128/JVI.79.24.15405-15416.2005
Overexpression of tumor necrosis factor alpha by a recombinant rabies virus attenuates replication in neurons and prevents lethal infection in mice
Abstract
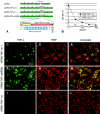
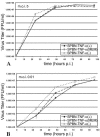
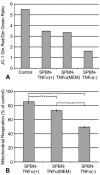
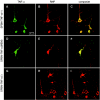
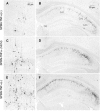
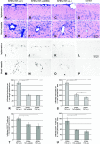
The effect of tumor necrosis factor alpha (TNF-alpha) on rabies virus (RV) infection of the mouse central nervous system (CNS) was studied, using recombinant RV engineered to express either soluble TNF-alpha [SPBN-TNF-alpha+] or insoluble membrane-bound TNF-alpha [SPBN-TNF-alpha(MEM)]. Growth curves derived from infections of mouse neuroblastoma NA cells revealed significantly less spread and production of SPBN-TNF-alpha+ than of SPBN-TNF-alpha(MEM) or SPBN-TNF-alpha-, which carries an inactivated TNF-alpha gene. The expression of soluble or membrane-bound TNF-alpha was not associated with increased cell death or induction of alpha/beta interferons. Brains of mice infected intranasally with SPBN-TNF-alpha+ showed significantly less virus spread than did mouse brains after SPBN-TNF-alpha- infection, and none of the SPBN-TNF-alpha+-infected mice succumbed to RV infection, whereas 80% of SPBN-TNF-alpha- -infected mice died. Reduced virus spread in SPBN-TNF-alpha+-infected mouse brains was paralleled by enhanced CNS inflammation, including T-cell infiltration and microglial activation. These data suggest that TNF-alpha exerts its protective activity in the brain directly through an as yet unknown antiviral mechanism and indirectly through the induction of inflammatory processes in the CNS.
Figures


Similar articles
-
Immune modulating effect by a phosphoprotein-deleted rabies virus vaccine vector expressing two copies of the rabies virus glycoprotein gene.Vaccine. 2008 Nov 25;26(50):6405-14. doi: 10.1016/j.vaccine.2008.08.069. Epub 2008 Sep 18. Vaccine. 2008. PMID: 18804506 Free PMC article.
-
Overexpression of cytochrome C by a recombinant rabies virus attenuates pathogenicity and enhances antiviral immunity.J Virol. 2001 Nov;75(22):10800-7. doi: 10.1128/JVI.75.22.10800-10807.2001. J Virol. 2001. PMID: 11602721 Free PMC article.
-
Role of virus-induced neuropeptides in the brain in the pathogenesis of rabies.Dev Biol (Basel). 2008;131:73-81. Dev Biol (Basel). 2008. PMID: 18634468
-
Replication strategies of rabies virus.Virus Res. 2005 Aug;111(2):120-31. doi: 10.1016/j.virusres.2005.04.004. Virus Res. 2005. PMID: 15885837 Review.
-
Canine adenovirus based rabies vaccines.Dev Biol (Basel). 2008;131:467-76. Dev Biol (Basel). 2008. PMID: 18634509 Review.
Cited by
-
Treat the "Untreatable" by a Photothermal Agent: Triggering Heat and Immunological Responses for Rabies Virus Inactivation.Adv Sci (Weinh). 2023 Jan;10(2):e2205461. doi: 10.1002/advs.202205461. Epub 2022 Nov 17. Adv Sci (Weinh). 2023. PMID: 36385484 Free PMC article.
-
TNF-overexpression in Borna disease virus-infected mouse brains triggers inflammatory reaction and epileptic seizures.PLoS One. 2012;7(7):e41476. doi: 10.1371/journal.pone.0041476. Epub 2012 Jul 25. PLoS One. 2012. PMID: 22848506 Free PMC article.
-
Characterisation of a Live-Attenuated Rabies Virus Expressing a Secreted scFv for the Treatment of Rabies.Viruses. 2023 Jul 31;15(8):1674. doi: 10.3390/v15081674. Viruses. 2023. PMID: 37632016 Free PMC article.
-
Rabies virus expressing dendritic cell-activating molecules enhances the innate and adaptive immune response to vaccination.J Virol. 2011 Feb;85(4):1634-44. doi: 10.1128/JVI.01552-10. Epub 2010 Nov 24. J Virol. 2011. PMID: 21106736 Free PMC article.
-
Lyssaviruses: current trends.Adv Virus Res. 2008;71:207-50. doi: 10.1016/S0065-3527(08)00005-5. Adv Virus Res. 2008. PMID: 18585530 Free PMC article. Review.
References
-
- Alberti, L., M. C. Thomachot, T. Bachelot, C. Menetrier-Caux, I. Puisieux, and J. Y. Blay. 2004. IL-6 as an intracrine growth factor for renal carcinoma cell lines. Int. J. Cancer 111:653-661. - PubMed
-
- Bette, M., O. Kaut, M. K. Schafer, and E. Weihe. 2003. Constitutive expression of p55TNFR mRNA and mitogen-specific up-regulation of TNF alpha and p75TNFR mRNA in mouse brain. J. Comp. Neurol. 465:417-430. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

