Characterisation of adherens and tight junctional molecules in normal animal larynx; determining a suitable model for studying molecular abnormalities in human laryngopharyngeal reflux
- PMID: 16311345
- PMCID: PMC1770809
- DOI: 10.1136/jcp.2004.016972
Characterisation of adherens and tight junctional molecules in normal animal larynx; determining a suitable model for studying molecular abnormalities in human laryngopharyngeal reflux
Abstract
Background: The disruption of intercellular junctions in the larynx is a pathological feature of laryngopharyngeal reflux (LPR). Good experimental models are necessary to gain greater insight into the molecular mechanisms and alterations that result from abnormal exposure of the laryngeal epithelium to acid refluxate.
Aims: To characterise laryngeal tissues from different species to determine the most suitable for use in experimental studies of LPR.
Methods: Human and non-human laryngeal tissues (mouse, rat, guinea pig, porcine, and rabbit) were studied. Histological characterisation was performed by light microscopy. The expression and subcellular localisation of adherens junctional molecules (E-cadherin and beta catenin) was evaluated by immunohistochemistry, and tight junction molecules (occludin and zonula occludens 1 (ZO-1)) by western blotting. The ultrastructural features of porcine and human tissue were assessed by electron microscopy.
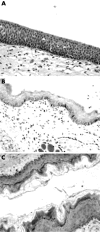
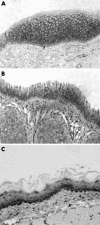
Results: Porcine tissue revealed both respiratory-type and stratified squamous epithelium, as seen in the human larynx. The expression and subcellular localisation of the E-cadherin-catenin complex was detected in all species except mouse and rat. The pattern of ZO-1 and occludin expression was preserved in all species.
Conclusion: The expression of intercellular junctional complexes in porcine epithelium is similar to that seen in humans. These results confirm the suitability of these species to study molecular mechanisms of LPR in an experimental system.
Figures




Similar articles
-
E-cadherin but not beta-catenin expression is decreased in laryngeal biopsies from patients with laryngopharyngeal reflux.Eur Arch Otorhinolaryngol. 2008 Aug;265(8):937-42. doi: 10.1007/s00405-007-0568-6. Epub 2008 Jan 5. Eur Arch Otorhinolaryngol. 2008. PMID: 18183411
-
Neutrophil transmigration in inflammatory bowel disease is associated with differential expression of epithelial intercellular junction proteins.Am J Pathol. 2001 Dec;159(6):2001-9. doi: 10.1016/S0002-9440(10)63051-9. Am J Pathol. 2001. PMID: 11733350 Free PMC article.
-
Laryngeal epithelial defenses against laryngopharyngeal reflux: investigations of E-cadherin, carbonic anhydrase isoenzyme III, and pepsin.Ann Otol Rhinol Laryngol. 2005 Dec;114(12):913-21. doi: 10.1177/000348940511401204. Ann Otol Rhinol Laryngol. 2005. PMID: 16425556
-
MicroRNA Regulation of Endothelial Junction Proteins and Clinical Consequence.Mediators Inflamm. 2016;2016:5078627. doi: 10.1155/2016/5078627. Epub 2016 Nov 24. Mediators Inflamm. 2016. PMID: 27999452 Free PMC article. Review.
-
Interplay between tight junctions & adherens junctions.Exp Cell Res. 2017 Sep 1;358(1):39-44. doi: 10.1016/j.yexcr.2017.03.061. Epub 2017 Mar 31. Exp Cell Res. 2017. PMID: 28372972 Free PMC article. Review.
Cited by
-
Human embryonic stem cell-derived epithelial cells in a novel in vitro model of vocal mucosa.Tissue Eng Part A. 2013 Oct;19(19-20):2233-41. doi: 10.1089/ten.TEA.2012.0744. Epub 2013 Jun 26. Tissue Eng Part A. 2013. PMID: 23672433 Free PMC article.
-
Vocal fold ion transport and mucin expression following acrolein exposure.J Membr Biol. 2014 May;247(5):441-50. doi: 10.1007/s00232-014-9651-2. Epub 2014 Mar 20. J Membr Biol. 2014. PMID: 24648011 Free PMC article.
-
Prolonged phonation impairs the integrity and barrier function of porcine vocal fold epithelium: a preliminary study.Eur Arch Otorhinolaryngol. 2018 Jun;275(6):1547-1556. doi: 10.1007/s00405-018-4973-9. Epub 2018 Apr 18. Eur Arch Otorhinolaryngol. 2018. PMID: 29671091
-
Hypertonic challenge to porcine vocal folds: effects on epithelial barrier function.Otolaryngol Head Neck Surg. 2010 Jan;142(1):79-84. doi: 10.1016/j.otohns.2009.09.011. Epub 2009 Nov 22. Otolaryngol Head Neck Surg. 2010. PMID: 20096227 Free PMC article.
-
Vocal fold injury models in rats: a literature review on techniques and methodology.J Med Life. 2022 Mar;15(3):336-343. doi: 10.25122/jml-2022-0032. J Med Life. 2022. PMID: 35449991 Free PMC article. Review.
References
-
- Amin MR, Postma GN, Johnson P, et al. Proton pump inhibitor resistance in the treatment of laryngopharyngeal reflux. Otolaryngol Head Neck Surg 2001;125:374–8. - PubMed
-
- Axford SE, Sharp N, Ross PE, et al. Cell biology of laryngeal epithelial defenses in health and disease: preliminary studies. Ann Otol Rhinol Laryngol 2001;110:1099–108. - PubMed
-
- Belafsky PC, Postma GN, Koufman JA. Laryngopharyngeal reflux symptoms improve before changes in physical findings. Laryngoscope 2001;111:979–81. - PubMed
-
- Koufman JA. Laryngopharyngeal reflux is different from classic gastroesophageal reflux disease. Ear Nose Throat J 2002;81 (suppl 2) :7–9. - PubMed
-
- Cohen JT, Bach KK, Postma GN, et al. Clinical manifestations of laryngopharyngeal reflux. Ear Nose Throat J 2002;81 (suppl 2) :19–23. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
