Complete remission of liver metastasis of pancreatic cancer under vaccination with a HLA-A2 restricted peptide derived from the universal tumor antigen survivin
- PMID: 16315030
- PMCID: PMC11031112
- DOI: 10.1007/s00262-005-0102-x
Complete remission of liver metastasis of pancreatic cancer under vaccination with a HLA-A2 restricted peptide derived from the universal tumor antigen survivin
Abstract
Purpose: As prognosis of advanced pancreatic cancer remains gloomy, novel therapeutic modalities have to be developed. Immunotherapy, which targets tumor-associated antigens of tumor cells or tumor stroma, is currently under investigation. As survivin is expressed by neoplastic and tumor endothelial cells, but rarely by normal cells, this antigen appears as an intriguing target molecule.
Methods: A 72-year old patient, suffering from pancreatic cancer refractory to gemcitabine therapy, received the survivin-based peptide vaccinations consisting of 100 mug of a modified HLA-A2 restricted survivin(96-104) epitope in Montanide(R). Each visit the patient was assessed for adverse events, quality of life and immunological response. Immuno-monitoring was performed by IFN-gamma-ELISPOT analysis of peripheral blood lymphocytes. Clinical outcome was evaluated by repetitive computed tomography.
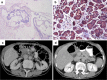
Results: Under vaccination with survivin peptides the patient initially underwent partial remission of liver metastasis which proceeded after 6 months into a complete remission with a duration of 8 months. Immunological monitoring revealed strong vaccine-induced immune-reactivity against survivin. Unfortunately, after the patient was weaned from vaccination in state of no evidence of disease, he developed recurrent disease.
Conclusion: T-cell responses against survivin-expressing cells of the tumor itself and tumor endothelium should impact tumor growth and metastasis. The presented patient with pancreatic cancer is the first example of a successful application of a survivin-based vaccination in the clinical setting. An ongoing phase I/II trial with HLA-A1, -A2 and -B35 restricted survivin peptides for patients with advanced cancer will provide further information towards this notion.
Figures

Similar articles
-
Immunologic response to the survivin-derived multi-epitope vaccine EMD640744 in patients with advanced solid tumors.Cancer Immunol Immunother. 2014 Apr;63(4):381-94. doi: 10.1007/s00262-013-1516-5. Epub 2014 Feb 2. Cancer Immunol Immunother. 2014. PMID: 24487961 Free PMC article. Clinical Trial.
-
Lack of toxicity of therapy-induced T cell responses against the universal tumour antigen survivin.Vaccine. 2005 Jan 4;23(7):884-9. doi: 10.1016/j.vaccine.2004.08.007. Vaccine. 2005. PMID: 15603888
-
Survivin-specific T-cell reactivity correlates with tumor response and patient survival: a phase-II peptide vaccination trial in metastatic melanoma.Cancer Immunol Immunother. 2012 Nov;61(11):2091-103. doi: 10.1007/s00262-012-1266-9. Epub 2012 May 8. Cancer Immunol Immunother. 2012. PMID: 22565484 Free PMC article. Clinical Trial.
-
Immune modulations during chemoimmunotherapy & novel vaccine strategies--in metastatic melanoma and non small-cell lung cancer.Dan Med J. 2013 Dec;60(12):B4774. Dan Med J. 2013. PMID: 24355457 Review.
-
Design of multi-epitope, analogue-based cancer vaccines.Expert Opin Biol Ther. 2003 Sep;3(6):985-93. doi: 10.1517/14712598.3.6.985. Expert Opin Biol Ther. 2003. PMID: 12943457 Review.
Cited by
-
BIRC5/Survivin as a target for glycolysis inhibition in high-stage neuroblastoma.Oncogene. 2016 Apr 21;35(16):2052-61. doi: 10.1038/onc.2015.264. Epub 2015 Jul 6. Oncogene. 2016. PMID: 26148234
-
Identification of HLA-A*0201-restricted cytotoxic T lymphocyte epitope from proliferating cell nuclear antigen.Tumour Biol. 2011 Feb;32(1):63-9. doi: 10.1007/s13277-010-0098-5. Epub 2010 Aug 16. Tumour Biol. 2011. PMID: 20711822
-
Generation of a novel transgenic mouse model for bioluminescent monitoring of survivin gene activity in vivo at various pathophysiological processes: survivin expression overlaps with stem cell markers.Am J Pathol. 2010 Apr;176(4):1629-38. doi: 10.2353/ajpath.2010.090414. Epub 2010 Feb 4. Am J Pathol. 2010. PMID: 20133811 Free PMC article.
-
Multidrug-resistance proteins are weak tumor associated antigens for colorectal carcinoma.BMC Immunol. 2011 Jul 10;12:38. doi: 10.1186/1471-2172-12-38. BMC Immunol. 2011. PMID: 21740599 Free PMC article.
-
Induction of HLA-DP4-restricted anti-survivin Th1 and Th2 responses using an artificial antigen-presenting cell.Clin Cancer Res. 2011 Aug 15;17(16):5392-401. doi: 10.1158/1078-0432.CCR-10-3083. Epub 2011 Jun 24. Clin Cancer Res. 2011. PMID: 21705450 Free PMC article.
References
-
- Andersen MH, Pedersen LO, Capeller B, Brocker EB, Becker JC, Straten PT. Spontaneous cytotoxic T-cell responses against survivin-derived MHC class I-restricted T-cell epitopes in situ as well as ex vivo in cancer patients. Cancer Res. 2001;61:5964–5968. - PubMed
-
- Berger TG, Haendle I, Schrama D, Luftl M, Bauer N, Pedersen LO, Schuler-Thurner B, Hohenberger W, Straten PT, Schuler G, Becker JC. Circulation and homing of melanoma-reactive T cells to both cutaneous and visceral metastases after vaccination with monocyte-derived dendritic cells. Int J Cancer. 2004;20(111):229–237. doi: 10.1002/ijc.20238. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

