Development of layer-specific axonal arborizations in mouse primary somatosensory cortex
- PMID: 16320250
- PMCID: PMC4651208
- DOI: 10.1002/cne.20754
Development of layer-specific axonal arborizations in mouse primary somatosensory cortex
Abstract
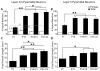
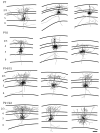
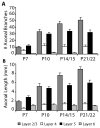
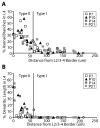
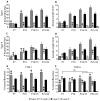
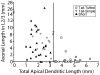
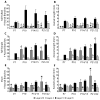
In the developing neocortex, pyramidal neurons use molecular cues to form axonal arbors selectively in the correct layers. Despite the utility of mice for molecular and genetic studies, little work has been done on the development of layer-specific axonal arborizations of pyramidal neurons in mice. We intracellularly labeled and reconstructed the axons of layer 2/3 and layer 5 pyramidal neurons in slices of primary somatosensory cortex from C57Bl6 mice on postnatal days 7-21. For all neurons studied, the development of the axonal arborizations in mice follows a pattern similar to that seen in other species; laminar specificity of the earliest axonal branches is similar to that of mature animals. At P7, pyramidal neurons are very simple, having only a main descending axon and few primary branches. Between P7 and P10, there is a large increase in the total number of axonal branches, and axons continue to increase in complexity and total length from P10 to P21. Unlike observations in ferrets, cats, and monkeys, two types of layer 2/3 pyramidal neurons are present in both mature and developing mice; cells in superficial layer 2/3 lack axonal arbors in layer 4, and cells close to the layer 4 border have substantial axonal arbors within layer 4. We also describe axonal and dendritic arborization patterns of three pyramidal cell types in layer 5. The axons of tall-tufted layer 5 pyramidal neurons arborize almost exclusively within deep layers while tall-simple, and short layer 5 pyramidal neurons also project axons to superficial layers.
Copyright 2005 Wiley-Liss, Inc
Figures




Similar articles
-
Development of axonal arbors of layer 6 pyramidal neurons in ferret primary visual cortex.J Comp Neurol. 1996 Dec 9;376(2):295-305. doi: 10.1002/(SICI)1096-9861(19961209)376:2<295::AID-CNE10>3.0.CO;2-L. J Comp Neurol. 1996. PMID: 8951644
-
Prenatal development of layer-specific local circuits in primary visual cortex of the macaque monkey.J Neurosci. 1998 Feb 15;18(4):1505-27. doi: 10.1523/JNEUROSCI.18-04-01505.1998. J Neurosci. 1998. PMID: 9454858 Free PMC article.
-
Laminar specificity of local circuits in barrel cortex of ephrin-A5 knockout mice.J Neurosci. 2000 Aug 1;20(15):RC88. doi: 10.1523/JNEUROSCI.20-15-j0005.2000. J Neurosci. 2000. PMID: 10899175 Free PMC article.
-
Columnar organization of dendrites and axons of single and synaptically coupled excitatory spiny neurons in layer 4 of the rat barrel cortex.J Neurosci. 2000 Jul 15;20(14):5300-11. doi: 10.1523/JNEUROSCI.20-14-05300.2000. J Neurosci. 2000. PMID: 10884314 Free PMC article.
-
Insights into the complex influence of 5-HT signaling on thalamocortical axonal system development.Eur J Neurosci. 2012 May;35(10):1563-72. doi: 10.1111/j.1460-9568.2012.8096.x. Eur J Neurosci. 2012. PMID: 22607002 Free PMC article. Review.
Cited by
-
The Anti-Inflammatory Agent Bindarit Attenuates the Impairment of Neural Development through Suppression of Microglial Activation in a Neonatal Hydrocephalus Mouse Model.J Neurosci. 2022 Mar 2;42(9):1820-1844. doi: 10.1523/JNEUROSCI.1160-21.2021. Epub 2022 Jan 6. J Neurosci. 2022. PMID: 34992132 Free PMC article.
-
Targeted approaches to delineate neuronal morphology during early development.Front Cell Neurosci. 2023 Oct 3;17:1259360. doi: 10.3389/fncel.2023.1259360. eCollection 2023. Front Cell Neurosci. 2023. PMID: 37854514 Free PMC article. Review.
-
Diverse Long-Range Axonal Projections of Excitatory Layer 2/3 Neurons in Mouse Barrel Cortex.Front Neuroanat. 2018 May 1;12:33. doi: 10.3389/fnana.2018.00033. eCollection 2018. Front Neuroanat. 2018. PMID: 29765308 Free PMC article.
-
Homeostasis of neuronal avalanches during postnatal cortex development in vitro.J Neurosci Methods. 2008 Apr 30;169(2):405-16. doi: 10.1016/j.jneumeth.2007.10.021. Epub 2007 Nov 7. J Neurosci Methods. 2008. PMID: 18082894 Free PMC article.
-
An Attempt at a Unified Theory of the Neocortical Microcircuit in Sensory Cortex.Front Neural Circuits. 2020 Jul 28;14:40. doi: 10.3389/fncir.2020.00040. eCollection 2020. Front Neural Circuits. 2020. PMID: 32848632 Free PMC article.
References
-
- Akemann W, Zhong YM, Ichinohe N, Rockland KS, Knopfel T. Transgenic mice expressing a fluorescent in vivo label in a distinct subpopulation of neocortical layer 5 pyramidal cells. J Comp Neurol. 2004;480:72–88. - PubMed
-
- Angevine JB, Sidman RL. Autoradiographic Study of Cell Migration during Histogenesis of Cerebral Cortex in the Mouse. Nature. 1961;192:766–768. - PubMed
-
- Annis CM, Robertson RT, O’Dowd DK. Aspects of early postnatal development of cortical neurons that proceed independently of normally present extrinsic influences. J Neurobiol. 1993;24:1460–1480. - PubMed
-
- Arimatsu Y, Ishida M. Distinct neuronal populations specified to form corticocortical and corticothalamic projections from layer VI of developing cerebral cortex. Neuroscience. 2002;114:1033–1045. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

