Solution structure of a novel tryptophan-rich peptide with bidirectional antimicrobial activity
- PMID: 16352849
- PMCID: PMC1317575
- DOI: 10.1128/JB.188.1.328-334.2006
Solution structure of a novel tryptophan-rich peptide with bidirectional antimicrobial activity
Abstract
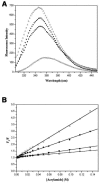
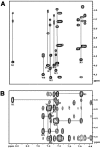
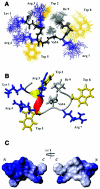
Trp-rich antimicrobial peptides play important roles in the host innate defense mechanisms of many plants, insects, and mammals. A new type of Trp-rich peptide, Ac-KWRRWVRWI-NH(2), designated Pac-525, was found to possess improved activity against both gram-positive and -negative bacteria. We have determined that the solution structures of Pac-525 bound to membrane-mimetic sodium dodecyl sulfate (SDS) micelles. The SDS micelle-bound structure of Pac-525 adopts an alpha-helical segment at residues Trp2, Arg3, and Arg4. The positively charged residues are clustered together to form a hydrophilic patch. The three hydrophobic residues Trp2, Val6, and Ile9 form a hydrophobic core. The surface electrostatic potential map indicates the three tryptophan indole rings are packed against the peptide backbone and form an amphipathic structure. Moreover, the reverse sequence of Pac-525, Ac-IWRVWRRWK-NH(2), designated Pac-525(rev), also demonstrates similar antimicrobial activity and structure in membrane-mimetic micelles and vesicles. A variety of biophysical and biochemical methods, including circular dichroism, fluorescence spectroscopy, and microcalorimetry, were used to show that Pac-525 interacted strongly with negatively charged phospholipid vesicles and induced efficient dye release from these vesicles, suggesting that the antimicrobial activity of Pac-525 may be due to interactions with bacterial membranes.
Figures
Similar articles
-
Solution structure of a novel D-naphthylalanine substituted peptide with potential antibacterial and antifungal activities.Biopolymers. 2007;88(5):738-45. doi: 10.1002/bip.20736. Biopolymers. 2007. PMID: 17410595
-
Rational design of tryptophan-rich antimicrobial peptides with enhanced antimicrobial activities and specificities.Chembiochem. 2010 Nov 2;11(16):2273-82. doi: 10.1002/cbic.201000372. Chembiochem. 2010. PMID: 20865718
-
Mechanism of antibacterial action of dermaseptin B2: interplay between helix-hinge-helix structure and membrane curvature strain.Biochemistry. 2009 Jan 20;48(2):313-27. doi: 10.1021/bi802025a. Biochemistry. 2009. PMID: 19113844
-
Proline-rich antimicrobial peptides: potential therapeutics against antibiotic-resistant bacteria.Amino Acids. 2014 Oct;46(10):2287-94. doi: 10.1007/s00726-014-1820-1. Epub 2014 Aug 21. Amino Acids. 2014. PMID: 25141976 Review.
-
Solution NMR studies of amphibian antimicrobial peptides: linking structure to function?Biochim Biophys Acta. 2009 Aug;1788(8):1639-55. doi: 10.1016/j.bbamem.2009.01.002. Epub 2009 Jan 15. Biochim Biophys Acta. 2009. PMID: 19272309 Review.
Cited by
-
How Insertion of a Single Tryptophan in the N-Terminus of a Cecropin A-Melittin Hybrid Peptide Changes Its Antimicrobial and Biophysical Profile.Membranes (Basel). 2021 Jan 12;11(1):48. doi: 10.3390/membranes11010048. Membranes (Basel). 2021. PMID: 33445476 Free PMC article.
-
Sarkosyl-Induced Helical Structure of an Antimicrobial Peptide GW-Q6 Plays an Essential Role in the Binding of Surface Receptor OprI in Pseudomonas aeruginosa.PLoS One. 2016 Oct 11;11(10):e0164597. doi: 10.1371/journal.pone.0164597. eCollection 2016. PLoS One. 2016. PMID: 27727309 Free PMC article.
-
Boosting salt resistance of short antimicrobial peptides.Antimicrob Agents Chemother. 2013 Aug;57(8):4050-2. doi: 10.1128/AAC.00252-13. Epub 2013 May 28. Antimicrob Agents Chemother. 2013. PMID: 23716061 Free PMC article.
-
Characterization of the structure-function relationship of a novel salt-resistant antimicrobial peptide, RR12.RSC Adv. 2020 Jun 22;10(40):23624-23631. doi: 10.1039/d0ra04299d. eCollection 2020 Jun 19. RSC Adv. 2020. PMID: 35517355 Free PMC article.
-
The pH sensitivity of histidine-containing lytic peptides.J Pept Sci. 2009 Nov;15(11):790-5. doi: 10.1002/psc.1180. J Pept Sci. 2009. PMID: 19787821 Free PMC article.
References
-
- Allen, T. M. (ed.). 1984. Calcein as a tool in liposome methodology. CRC Press, Boca Raton, Fla.
-
- Andreu, D., and L. Rivas. 1998. Animal antimicrobial peptides: an overview. Biopolymers 47:415-433. - PubMed
-
- Blondelle, S., and K. Lohner. 2000. Combinatorial libraries: a tool to design antimicrobial and antifungal peptide analogues having lytic specificities for structure-activity relationship studies. Biopolymers 55:74-87. - PubMed
-
- Brock, T. D. 1974. Biology of microorganisms, 2nd ed. Prentice-Hall Inc., Englewood Cliffs, N.J.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

