KLF4 and PCNA identify stages of tumor initiation in a conditional model of cutaneous squamous epithelial neoplasia
- PMID: 16357510
- PMCID: PMC1361751
- DOI: 10.4161/cbt.4.12.2355
KLF4 and PCNA identify stages of tumor initiation in a conditional model of cutaneous squamous epithelial neoplasia
Abstract
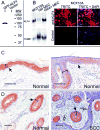
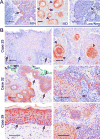
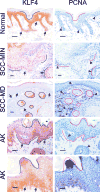
KLF4 is induced upon growth-arrest in vitro and during epithelial maturation in vivo, and is essential for proper cell fate specification of post-mitotic cells. In spite of a normal role in post-mitotic cells, expression is upregulated and constitutive in certain tumor types. KLF4 functions as an oncogene in vitro, and enforced expression in basal cells of mouse skin rapidly induces lesions similar to hyperplasia, dysplasia and squamous cell carcinoma (SCC). Here we used conditional expression to characterize early steps in KLF4-mediated tumor initiation. In contrast to SCC-like lesions that result when using a conditional, keratin 14 promoter-dependent strategy, lower conditional expression achieved using a MMTV promoter induced only epidermal cycling within morphologically normal skin, a process we termed occult cell turnover. Surprisingly, KLF4-induced hyperplastic lesions showed increased transgene-derived mRNA and protein in maturing, PCNA-negative cells, a property of endogenous KLF4. In contrast, hyperplastic lesions induced by GLI1, a control, showed uniform transgene expression. In KLF4-induced dysplasia and SCC the complementarity of KLF4 and PCNA was replaced by concordance of the two proteins. These studies show that KLF4 transcripts are normally suppressed in cycling cells in a promoter-independent fashion, consistent with a post-transcriptional control, and reveal loss of this control in the transition from hyperplasia to dysplasia. Like the mouse tumors, human cutaneous SCCs and adjacent dysplasias frequently showed maturation-independence of KLF4, with co-expression of KLF4 and PCNA. A smaller subset of human SCCs showed complementarity of KLF4 and PCNA, similar to hyperplastic mouse skin. The results identify parallels between a mouse model and human primary tumors, and show that successive increases of KLF4 in the nuclei of basal keratinocytes leads to occult cell turnover followed by hyperplasia, dysplasia, and invasive SCC.
Figures



Similar articles
-
KLF4 suppresses the tumor activity of cutaneous squamous cell carcinoma (SCC) cells via the regulation of SMAD signaling and SOX2 expression.Biochem Biophys Res Commun. 2019 Sep 3;516(4):1110-1115. doi: 10.1016/j.bbrc.2019.07.011. Epub 2019 Jul 6. Biochem Biophys Res Commun. 2019. PMID: 31284949
-
Deficiency of the Kruppel-like factor KLF4 correlates with increased cell proliferation and enhanced skin tumorigenesis.Carcinogenesis. 2012 Jun;33(6):1239-46. doi: 10.1093/carcin/bgs143. Epub 2012 Apr 9. Carcinogenesis. 2012. PMID: 22491752 Free PMC article.
-
Prevention of KLF4-mediated tumor initiation and malignant transformation by UAB30 rexinoid.Cancer Biol Ther. 2009 Feb;8(3):289-98. doi: 10.4161/cbt.8.3.7486. Epub 2009 Feb 21. Cancer Biol Ther. 2009. PMID: 19197145 Free PMC article.
-
Alterations in epidermal biochemistry as a consequence of stage-specific genetic changes in skin carcinogenesis.Environ Health Perspect. 1991 Jun;93:3-10. doi: 10.1289/ehp.91933. Environ Health Perspect. 1991. PMID: 1773799 Free PMC article. Review.
-
UV-Induced Molecular Signaling Differences in Melanoma and Non-melanoma Skin Cancer.Adv Exp Med Biol. 2017;996:27-40. doi: 10.1007/978-3-319-56017-5_3. Adv Exp Med Biol. 2017. PMID: 29124688 Review.
Cited by
-
KLF4 downregulates hTERT expression and telomerase activity to inhibit lung carcinoma growth.Oncotarget. 2016 Aug 16;7(33):52870-52887. doi: 10.18632/oncotarget.9141. Oncotarget. 2016. PMID: 27153563 Free PMC article.
-
Recent developments in targeting breast cancer stem cells (BCSCs): a descriptive review of therapeutic strategies and emerging therapies.Med Oncol. 2024 Apr 9;41(5):112. doi: 10.1007/s12032-024-02347-z. Med Oncol. 2024. PMID: 38592510 Review.
-
Leptin stimulates synaptogenesis in hippocampal neurons via KLF4 and SOCS3 inhibition of STAT3 signaling.Mol Cell Neurosci. 2020 Jul;106:103500. doi: 10.1016/j.mcn.2020.103500. Epub 2020 May 11. Mol Cell Neurosci. 2020. PMID: 32438059 Free PMC article.
-
A novel KLF4/LDHA signaling pathway regulates aerobic glycolysis in and progression of pancreatic cancer.Clin Cancer Res. 2014 Aug 15;20(16):4370-80. doi: 10.1158/1078-0432.CCR-14-0186. Epub 2014 Jun 19. Clin Cancer Res. 2014. PMID: 24947925 Free PMC article.
-
Expression and Function of Kruppel Like-Factors (KLF) in Carcinogenesis.Curr Genomics. 2009 Aug;10(5):353-60. doi: 10.2174/138920209788921010. Curr Genomics. 2009. PMID: 20119532 Free PMC article.
References
-
- Taipale J, Beachy PA. The Hedgehog and Wnt signaling pathways in cancer. Nature. 2001;411:349–54. - PubMed
-
- Vogelstein B, Kinzler KW. Cancer genes and the pathways they control. Nat Med. 2004;10:789–99. - PubMed
-
- Nusse R. Wnts and Hedgehogs: lipid-modified proteins and similarities in signaling mechanisms at the cell surface. Development. 2003;130:5297–305. - PubMed
-
- Pires-daSilva A, Sommer RJ. The evolution of signalling pathways in animal development. Nat Rev Genet. 2003;4:39–49. - PubMed
-
- Foster KW, Ren S, Louro ID, Lobo-Ruppert SM, McKie-Bell P, Grizzle W, Hayes MR, Broker TR, Chow LT, Ruppert JM. Oncogene expression cloning by retroviral transduction of adenovirus E1a-immortalized rat kidney RK3E cells: transformation of a host with epithelial features by c-MYC and the zinc finger protein GKLF. Cell Growth Differ. 1999;10:423–34. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous
