Determination of theophylline concentration in serum by chemiluminescent immunoassay
- PMID: 16358370
- PMCID: PMC1390635
- DOI: 10.1631/jzus.2005.B1148
Determination of theophylline concentration in serum by chemiluminescent immunoassay
Abstract
Objective: This study aimed to establish chemiluminescent immunoassay (CLIA) for quantitative determination of theophylline levels in human serum.
Methods: To measure the concentration of theophylline (n=122) and evaluate the assay.
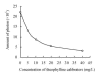
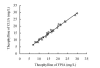
Results: The linear range of the CLIA method was 0.51-40 mg/L (Y=1.02X+0.44, r=0.995). The intra and inter CV (coefficient variance) of CLIA were 3.20% and 3.57%, respectively. The average recovery rate was 102.3%. This method was free from interference by brilirubin (<200 micromol/L), hemoglobin (<10 g/L), and triglycerides (<15 mmol/L).
Conclusion: This method is simple, convenient and precise for clinical pharmacokinetics study of theophylline.
Figures
Similar articles
-
Comparison of the automated AxSYM and ADVIA centaur immunoassays for homocysteine determination.Clin Lab. 2004;50(3-4):175-80. Clin Lab. 2004. PMID: 15074472
-
Determination of estradiol in human serum using magnetic particles-based chemiluminescence immunoassay.Anal Chim Acta. 2008 Oct 10;627(2):277-84. doi: 10.1016/j.aca.2008.08.020. Epub 2008 Aug 28. Anal Chim Acta. 2008. PMID: 18809084
-
Rapid immunoassay for the determination of glial fibrillary acidic protein (GFAP) in serum.Clin Chim Acta. 2006 Apr;366(1-2):336-40. doi: 10.1016/j.cca.2005.11.017. Epub 2006 Jan 10. Clin Chim Acta. 2006. PMID: 16405937
-
Specific methods for theophylline assay in biological samples.Eur J Respir Dis Suppl. 1980;109:45-53. Eur J Respir Dis Suppl. 1980. PMID: 7002578 Review.
-
[Calcitonin assay].Nihon Rinsho. 2005 Oct;63 Suppl 10:194-7. Nihon Rinsho. 2005. PMID: 16279629 Review. Japanese. No abstract available.
Cited by
-
A Disposable Sensor Chip Using a Paste Electrode with Surface-Imprinted Graphite Particles for Rapid and Reagentless Monitoring of Theophylline.Molecules. 2022 Apr 11;27(8):2456. doi: 10.3390/molecules27082456. Molecules. 2022. PMID: 35458653 Free PMC article.
-
An electrochemical sensor based on a modified glassy carbon electrode for detection of epinephrine in the presence of theophylline.ADMET DMPK. 2024 Apr 15;12(2):391-402. doi: 10.5599/admet.2082. eCollection 2024. ADMET DMPK. 2024. PMID: 38720927 Free PMC article.
-
Nano optical and electrochemical sensors and biosensors for detection of narrow therapeutic index drugs.Mikrochim Acta. 2021 Nov 6;188(12):411. doi: 10.1007/s00604-021-05003-9. Mikrochim Acta. 2021. PMID: 34741213 Review.
-
Development of an electrochemical immunoassay for detection of gatifloxacin in swine urine.J Zhejiang Univ Sci B. 2012 Feb;13(2):118-25. doi: 10.1631/jzus.B1100073. J Zhejiang Univ Sci B. 2012. PMID: 22302425 Free PMC article.
References
-
- Amal L, Bergmann P. Evaluation of a chemiluminescence immunoassay for the determination of intact parathyroid hormone using the ADVIA Centaur. Clin Lab. 2004;50(11-12):695–702. - PubMed
-
- Gupta SV, McGowen RM, Callewaert DM, Brown TR, Li Y, Sarkar FH. Quantitative chemiluminescent immunoassay for NF-kappaB-DNA binding activity. J Immunoassay Immunochem. 2005;26(2):125–143. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical