Translational control in positive strand RNA plant viruses
- PMID: 16364749
- PMCID: PMC1847782
- DOI: 10.1016/j.virol.2005.09.031
Translational control in positive strand RNA plant viruses
Abstract
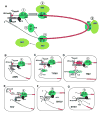
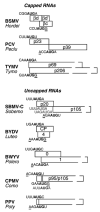
The great variety of genome organizations means that most plant positive strand viral RNAs differ from the standard 5'-cap/3'-poly(A) structure of eukaryotic mRNAs. The cap and poly(A) tail recruit initiation factors that support the formation of a closed loop mRNA conformation, the state in which translation initiation is most efficient. We review the diverse array of cis-acting sequences present in viral mRNAs that compensate for the absence of a cap, poly(A) tail, or both. We also discuss the cis-acting sequences that control translation strategies that both amplify the coding potential of a genome and regulate the accumulations of viral gene products. Such strategies include leaky scanning initiation of translation of overlapping open reading frames, stop codon readthrough, and ribosomal frameshifting. Finally, future directions for research on the translation of plant positive strand viruses are discussed.
Figures

Similar articles
-
Translation of Plant RNA Viruses.Viruses. 2021 Dec 13;13(12):2499. doi: 10.3390/v13122499. Viruses. 2021. PMID: 34960768 Free PMC article. Review.
-
3' cap-independent translation enhancers of plant viruses.Annu Rev Microbiol. 2013;67:21-42. doi: 10.1146/annurev-micro-092412-155609. Epub 2013 May 13. Annu Rev Microbiol. 2013. PMID: 23682606 Free PMC article. Review.
-
3' Cap-independent translation enhancers of positive-strand RNA plant viruses.Curr Opin Virol. 2011 Nov;1(5):373-80. doi: 10.1016/j.coviro.2011.10.002. Epub 2011 Oct 24. Curr Opin Virol. 2011. PMID: 22440838 Review.
-
In vitro translation of plant viral RNA.Curr Protoc Microbiol. 2006 Jun;Chapter 16:16K.1.1-16K.1.13. doi: 10.1002/9780471729259.mc16k01s01. Curr Protoc Microbiol. 2006. PMID: 18770587
-
Short 5' Untranslated Region Enables Optimal Translation of Plant Virus Tricistronic RNA via Leaky Scanning.J Virol. 2022 Apr 13;96(7):e0214421. doi: 10.1128/jvi.02144-21. Epub 2022 Mar 9. J Virol. 2022. PMID: 35262378 Free PMC article.
Cited by
-
Crinivirus replication and host interactions.Front Microbiol. 2013 May 20;4:99. doi: 10.3389/fmicb.2013.00099. eCollection 2013. Front Microbiol. 2013. PMID: 23730299 Free PMC article.
-
Tombusvirus recruitment of host translational machinery via the 3' UTR.RNA. 2010 Jul;16(7):1402-19. doi: 10.1261/rna.2135210. Epub 2010 May 27. RNA. 2010. PMID: 20507975 Free PMC article.
-
Viral condensates formed by Pea enation mosaic virus 2 sequester ribosomal components and suppress translation.Virology. 2025 Jan;601:110301. doi: 10.1016/j.virol.2024.110301. Epub 2024 Nov 13. Virology. 2025. PMID: 39549315
-
A ribosome-binding, 3' translational enhancer has a T-shaped structure and engages in a long-distance RNA-RNA interaction.J Virol. 2012 Sep;86(18):9828-42. doi: 10.1128/JVI.00677-12. Epub 2012 Jul 3. J Virol. 2012. PMID: 22761367 Free PMC article.
-
Augmented genetic decoding: global, local and temporal alterations of decoding processes and codon meaning.Nat Rev Genet. 2015 Sep;16(9):517-29. doi: 10.1038/nrg3963. Epub 2015 Aug 11. Nat Rev Genet. 2015. PMID: 26260261 Review.
References
-
- Aranda M, Maule A. Virus-induced host gene shutoff in animals and plants. Virology. 1998;243:261–267. - PubMed
-
- Baranov PV, Gesteland RF, Atkins JF. Recording: translational bifurcations in gene expression. Gene. 2002;286:187–201. - PubMed
-
- Barends S, Bink HH, van den Worm SH, Pleij CW, Kraal B. Entrapping ribosomes for viral translation: tRNA mimicry as a molecular Trojan horse. Cell. 2003;112:123–129. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

