Synantocytes: the fifth element
- PMID: 16367797
- PMCID: PMC1571581
- DOI: 10.1111/j.1469-7580.2005.00458.x
Synantocytes: the fifth element
Abstract
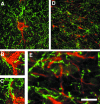
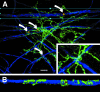
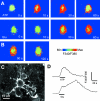
Classic studies have recognized neurons and three glial elements in the central nervous system (CNS) - astrocytes, oligodendrocytes and microglia. The identification of novel glia that specifically express the NG2 chondroitin sulphate proteoglycan (CSPG) raises the possibility of a fifth element. Until recently, all NG2-expressing glia were considered to be oligodendrocyte precursor cells (OPCs) that persist in the adult CNS to generate oligodendrocytes throughout life. However, this narrow view of the function of 'NG2-glia' is being challenged. The majority of NG2-expressing glia in the adult CNS are a distinct class of cells that we have called 'synantocytes' (from the Greek synanto for contact). Synantocytes are stellate cells, with large process arborizations, and are exquisitely related to neurons. Individual cells traverse white and grey matter and form multiple contacts with neurons, astrocytes, oligodendrocytes and myelin. Synantocytes are an integral component of the 'tetrapartite' synapse, and provide a potential integrative neuron-glial communications pathway. Neuronal activity, glutamate and adenosine triphosphate (ATP) act on synantocyte receptors and evoke raised intracellular calcium. It remains to be seen whether this serves a physiological function, but synantocytes may be specialized to monitor signals from neurons and glia, and to respond to changes in the integrity of the CNS via their specific contacts and ion channel and receptor profiles. The general consequences of synantocyte activation are proliferation and phenotypic changes, resulting in glial scar formation, or regeneration of oligodendrocytes, and possibly neurons.
Figures




References
-
- Araque A, Parpura V, Sanzgiri RP, Haydon PG. Tripartite synapses: glia, the unacknowledged partner. Trends Neurosci. 1999;22:208–215. - PubMed
-
- Barres BA, Koroshetz WJ, Swartz KJ, Chun LL, Corey DP. Ion channel expression by white matter glia: the O-2A glial progenitor cell. Neuron. 1990;4:507–524. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

