NMDA receptors are expressed in oligodendrocytes and activated in ischaemia
- PMID: 16372011
- PMCID: PMC1416283
- DOI: 10.1038/nature04302
NMDA receptors are expressed in oligodendrocytes and activated in ischaemia
Abstract
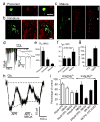
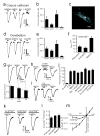
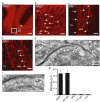
Glutamate-mediated damage to oligodendrocytes contributes to mental or physical impairment in periventricular leukomalacia (pre- or perinatal white matter injury leading to cerebral palsy), spinal cord injury, multiple sclerosis and stroke. Unlike neurons, white matter oligodendrocytes reportedly lack NMDA (N-methyl-d-aspartate) receptors. It is believed that glutamate damages oligodendrocytes, especially their precursor cells, by acting on calcium-permeable AMPA (alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid)/kainate receptors alone or by reversing cystine-glutamate exchange and depriving cells of antioxidant protection. Here we show that precursor, immature and mature oligodendrocytes in the white matter of the cerebellum and corpus callosum exhibit NMDA-evoked currents, mediated by receptors that are blocked only weakly by Mg2+ and that may contain NR1, NR2C and NR3 NMDA receptor subunits. NMDA receptors are present in the myelinating processes of oligodendrocytes, where the small intracellular space could lead to a large rise in intracellular ion concentration in response to NMDA receptor activation. Simulating ischaemia led to development of an inward current in oligodendrocytes, which was partly mediated by NMDA receptors. These results point to NMDA receptors of unusual subunit composition as a potential therapeutic target for preventing white matter damage in a variety of diseases.
Figures

Similar articles
-
NMDA receptors mediate calcium accumulation in myelin during chemical ischaemia.Nature. 2006 Feb 23;439(7079):988-92. doi: 10.1038/nature04474. Epub 2005 Dec 21. Nature. 2006. PMID: 16372019
-
The receptor subunits generating NMDA receptor mediated currents in oligodendrocytes.J Physiol. 2010 Sep 15;588(Pt 18):3403-14. doi: 10.1113/jphysiol.2010.195503. Epub 2010 Jul 26. J Physiol. 2010. PMID: 20660562 Free PMC article.
-
Testing NMDA receptor block as a therapeutic strategy for reducing ischaemic damage to CNS white matter.Glia. 2008 Jan 15;56(2):233-40. doi: 10.1002/glia.20608. Glia. 2008. PMID: 18046734 Free PMC article.
-
NMDA receptors expressed in oligodendrocytes.Bioessays. 2006 May;28(5):460-4. doi: 10.1002/bies.20402. Bioessays. 2006. PMID: 16615088 Review.
-
Age-specific localization of NMDA receptors on oligodendrocytes dictates axon function recovery after ischemia.Neuropharmacology. 2016 Nov;110(Pt B):626-632. doi: 10.1016/j.neuropharm.2015.09.015. Epub 2015 Sep 25. Neuropharmacology. 2016. PMID: 26407763 Free PMC article. Review.
Cited by
-
Olig2/Plp-positive progenitor cells give rise to Bergmann glia in the cerebellum.Cell Death Dis. 2013 Mar 14;4(3):e546. doi: 10.1038/cddis.2013.74. Cell Death Dis. 2013. PMID: 23492777 Free PMC article.
-
Selective Vulnerabilities of N-methyl-D-aspartate (NMDA) Receptors During Brain Aging.Front Aging Neurosci. 2010 Mar 19;2:11. doi: 10.3389/fnagi.2010.00011. eCollection 2010. Front Aging Neurosci. 2010. PMID: 20552049 Free PMC article.
-
Electrical and synaptic integration of glioma into neural circuits.Nature. 2019 Sep;573(7775):539-545. doi: 10.1038/s41586-019-1563-y. Epub 2019 Sep 18. Nature. 2019. PMID: 31534222 Free PMC article.
-
Regulated release of BDNF by cortical oligodendrocytes is mediated through metabotropic glutamate receptors and the PLC pathway.ASN Neuro. 2009 Apr 14;1(1):e00001. doi: 10.1042/AN20090006. ASN Neuro. 2009. PMID: 19570026 Free PMC article.
-
Piezo1-mediated spontaneous calcium transients in satellite glia impact dorsal root ganglia development.PLoS Biol. 2023 Sep 25;21(9):e3002319. doi: 10.1371/journal.pbio.3002319. eCollection 2023 Sep. PLoS Biol. 2023. PMID: 37747915 Free PMC article.
References
-
- Volpe JJ. Neurobiology of periventricular leukomalacia in the premature infant. Pediatr Res. 2001;50:553–562. - PubMed
-
- Stys PK. White matter injury mechanisms. Curr Mol Med. 2004;4:113–130. - PubMed
-
- Matute C, Alberdi E, Domercq M, Perez-Cerda F, Perez-Samartin A, Sanchez-Gomez MV. The link between excitotoxic oligodendroglial death and demyelinating diseases. Trends Neurosci. 2001;24:224–230. - PubMed
-
- Dewar D, Underhill SM, Goldberg MP. Oligodendrocytes and ischemic brain injury. J Cereb Blood Flow Metab. 2003;23:263–274. - PubMed
-
- Choi DW. Glutamate neurotoxicity and diseases of the nervous system. Neuron. 1988;1:623–634. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

