Erythropoietin requires NF-kappaB and its nuclear translocation to prevent early and late apoptotic neuronal injury during beta-amyloid toxicity
- PMID: 16375720
- PMCID: PMC1986681
- DOI: 10.2174/156720205774962683
Erythropoietin requires NF-kappaB and its nuclear translocation to prevent early and late apoptotic neuronal injury during beta-amyloid toxicity
Abstract
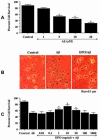
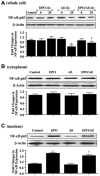
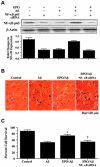
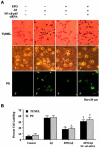
No longer considered exclusive for the function of the hematopoietic system, erythropoietin (EPO) is now considered as a viable agent to address central nervous system injury in a variety of cellular systems that involve neuronal, vascular, and inflammatory cells. Yet, it remains unclear whether the protective capacity of EPO may be effective for chronic neurodegenerative disorders such as Alzheimer's disease (AD) that involve beta-amyloid (Abeta) apoptotic injury to hippocampal neurons. We therefore investigated whether EPO could prevent both early and late apoptotic injury during Abeta exposure in primary hippocampal neurons and assessed potential cellular pathways responsible for this protection. Primary hippocampal neuronal injury was evaluated by trypan blue dye exclusion, DNA fragmentation, membrane phosphatidylserine (PS) exposure, and nuclear factor-kappaB (NF-kappaB) expression with subcellular translocation. We show that EPO, in a concentration specific manner, is able to prevent the loss of both apoptotic genomic DNA integrity and cellular membrane asymmetry during Abeta exposure. This blockade of Abeta generated neuronal apoptosis by EPO is both necessary and sufficient, since protection by EPO is completely abolished by co-treatment with an anti-EPO neutralizing antibody. Furthermore, neuroprotection by EPO is closely linked to the expression of NF-kappaB p65 by preventing the degradation of this protein by Abeta and fostering the subcellular translocation of NF-kappaB p65 from the cytoplasm to the nucleus to allow the initiation of an anti-apoptotic program. In addition, EPO intimately relies upon NF-kappaB p65 to promote neuronal survival, since gene silencing of NF-kappaB p65 by RNA interference removes the protective capacity of EPO during Abeta exposure. Our work illustrates that EPO is an effective entity at the neuronal cellular level against Abeta toxicity and requires the close modulation of the NF-kappaB p65 pathway, suggesting that either EPO or NF-kappaB may be used as future potential therapeutic strategies for the management of chronic neurodegenerative disorders, such as AD.
Figures


References
-
- Bernaudin M, Marti HH, Roussel S, Divoux D, Nouvelot A, MacKenzie ET, Petit E. A potential role for erythropoietin in focal permanent cerebral ischemia in mice. J Cereb Blood Flow Metab. 1999;19(6):643–51. - PubMed
-
- Bittorf T, Buchse T, Sasse T, Jaster R, Brock J. Activation of the transcription factor NF-kappaB by the erythropoietin receptor: structural requirements and biological significance. Cell Signal. 2001;13(9):673–81. - PubMed
-
- Carvalho G, Lefaucheur C, Cherbonnier C, Metivier D, Chapel A, Pallardy M, Bourgeade MF, Charpentier B, Hirsch F, Kroemer G. Chemosensitization by erythropoietin through inhibition of the NF-kappaB rescue pathway. Oncogene. 2005;24(5):737–45. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
