Bcl-2 differentially regulates Ca2+ signals according to the strength of T cell receptor activation
- PMID: 16391001
- PMCID: PMC2063540
- DOI: 10.1083/jcb.200506189
Bcl-2 differentially regulates Ca2+ signals according to the strength of T cell receptor activation
Abstract
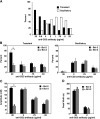
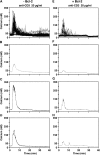
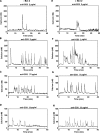
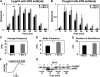
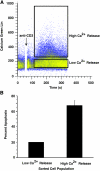
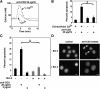
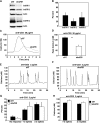
To investigate the effect of Bcl-2 on Ca2+ signaling in T cells, we continuously monitored Ca2+ concentration in Bcl-2-positive and -negative clones of the WEHI7.2 T cell line after T cell receptor (TCR) activation by anti-CD3 antibody. In Bcl-2-negative cells, high concentrations of anti-CD3 antibody induced a transient Ca2+ elevation, triggering apoptosis. In contrast, low concentrations of anti-CD3 antibody induced Ca2+ oscillations, activating the nuclear factor of activated T cells (NFAT), a prosurvival transcription factor. Bcl-2 blocked the transient Ca2+ elevation induced by high anti-CD3, thereby inhibiting apoptosis, but did not inhibit Ca2+ oscillations and NFAT activation induced by low anti-CD3. Reduction in the level of all three inositol 1,4,5-trisphosphate (InsP(3)) receptor subtypes by small interfering RNA inhibited the Ca2+ elevation induced by high but not low anti-CD3, suggesting that Ca2+ responses to high and low anti-CD3 may have different requirements for the InsP(3) receptor. Therefore, Bcl-2 selectively inhibits proapoptotic Ca2+ elevation induced by strong TCR activation without hindering prosurvival Ca2+ signals induced by weak TCR activation.
Figures

Similar articles
-
Bcl-2 functionally interacts with inositol 1,4,5-trisphosphate receptors to regulate calcium release from the ER in response to inositol 1,4,5-trisphosphate.J Cell Biol. 2004 Jul 19;166(2):193-203. doi: 10.1083/jcb.200309146. J Cell Biol. 2004. PMID: 15263017 Free PMC article.
-
V7 (CD101) ligation inhibits TCR/CD3-induced IL-2 production by blocking Ca2+ flux and nuclear factor of activated T cell nuclear translocation.J Immunol. 1998 Jul 1;161(1):209-17. J Immunol. 1998. PMID: 9647226
-
T-cell-receptor signalling in inositol 1,4,5-trisphosphate receptor (IP3R) type-1-deficient mice: is IP3R type 1 essential for T-cell-receptor signalling?Biochem J. 1998 Aug 1;333 ( Pt 3)(Pt 3):615-9. doi: 10.1042/bj3330615. Biochem J. 1998. PMID: 9677320 Free PMC article.
-
[Role of inositol triphosphate in the formation of the calcium signal in T-lymphocytes].Ukr Biokhim Zh (1978). 1998 Jul-Aug;70(4):3-15. Ukr Biokhim Zh (1978). 1998. PMID: 9848195 Review. Russian.
-
Live free or die: an immature T cell decision encoded in distinct Bcl-2 sensitive and insensitive Ca2+ signals.Cell Cycle. 2006 Jun;5(11):1171-4. doi: 10.4161/cc.5.11.2778. Epub 2006 Jun 1. Cell Cycle. 2006. PMID: 16721059 Review.
Cited by
-
Distinct Cellular Calcium Metabolism in Radiation-sensitive RKO Human Colorectal Cancer Cells.Korean J Physiol Pharmacol. 2014 Dec;18(6):509-16. doi: 10.4196/kjpp.2014.18.6.509. Epub 2014 Dec 30. Korean J Physiol Pharmacol. 2014. PMID: 25598666 Free PMC article.
-
The trans-membrane domain of Bcl-2α, but not its hydrophobic cleft, is a critical determinant for efficient IP3 receptor inhibition.Oncotarget. 2016 Aug 23;7(34):55704-55720. doi: 10.18632/oncotarget.11005. Oncotarget. 2016. PMID: 27494888 Free PMC article.
-
A Novel Peptide that Disrupts the Lck-IP3R Protein-Protein Interaction Induces Widespread Cell Death in Leukemia and Lymphoma.Arch Microbiol Immunol. 2023;7(3):165-177. doi: 10.26502/ami.936500114. Epub 2023 Aug 30. Arch Microbiol Immunol. 2023. PMID: 37829571 Free PMC article.
-
The correlation between mitochondria-associated endoplasmic reticulum membranes (MAMs) and Ca2+ transport in the pathogenesis of diseases.Acta Pharmacol Sin. 2025 Feb;46(2):271-291. doi: 10.1038/s41401-024-01359-9. Epub 2024 Aug 8. Acta Pharmacol Sin. 2025. PMID: 39117969 Review.
-
The proapoptotic factors Bax and Bak regulate T Cell proliferation through control of endoplasmic reticulum Ca(2+) homeostasis.Immunity. 2007 Aug;27(2):268-80. doi: 10.1016/j.immuni.2007.05.023. Epub 2007 Aug 9. Immunity. 2007. PMID: 17692540 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

