New rfp- and pES213-derived tools for analyzing symbiotic Vibrio fischeri reveal patterns of infection and lux expression in situ
- PMID: 16391121
- PMCID: PMC1352280
- DOI: 10.1128/AEM.72.1.802-810.2006
New rfp- and pES213-derived tools for analyzing symbiotic Vibrio fischeri reveal patterns of infection and lux expression in situ
Abstract
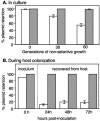
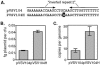
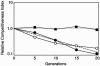
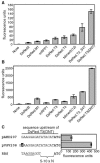
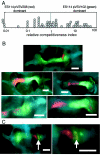
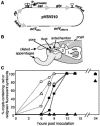
Genetically altered or tagged Vibrio fischeri strains can be observed in association with their mutualistic host Euprymna scolopes, providing powerful experimental approaches for studying this symbiosis. Two limitations to such in situ analyses are the lack of suitably stable plasmids and the need for a fluorescent tag that can be used in tandem with green fluorescent protein (GFP). Vectors previously used in V. fischeri contain the p15A replication origin; however, we found that this replicon is not stable during growth in the host and is retained by fewer than 20% of symbionts within a day after infection. In contrast, derivatives of V. fischeri plasmid pES213 were retained by approximately 99% of symbionts even 3 days after infection. We therefore constructed pES213-derived shuttle vectors with a variety of selectable and visual markers. To include a visual tag that can be used in conjunction with GFP, we compared seven variants of the DsRed2 red fluorescent protein (RFP): mRFP1, tdimer2(12), DsRed.T3, DsRed.T4, DsRed.M1, DsRed.T3_S4T, and DsRed.T3(DNT). The last variant was brightest, displaying >20-fold more fluorescence than DsRed2 in V. fischeri. RFP expression did not detectably affect the fitness of V. fischeri, and cells were readily visualized in combination with GFP-expressing cells in mixed infections. Interestingly, even when inocula were dense enough that most E. scolopes hatchlings were infected by two strains, there was little mixing of the strains in the light organ crypts. We also used constitutive RFP in combination with the luxICDABEG promoter driving expression of GFP to visualize the spatial and temporal induction of this bioluminescence operon during symbiotic infection. Our results demonstrate the utility of pES213-based vectors and RFP for in situ experimental approaches in studies of the V. fischeri-E. scolopes symbiosis.
Figures
Similar articles
-
Characterization of htrB and msbB mutants of the light organ symbiont Vibrio fischeri.Appl Environ Microbiol. 2008 Feb;74(3):633-44. doi: 10.1128/AEM.02138-07. Epub 2007 Dec 7. Appl Environ Microbiol. 2008. PMID: 18065606 Free PMC article.
-
Intraspecific Competition Impacts Vibrio fischeri Strain Diversity during Initial Colonization of the Squid Light Organ.Appl Environ Microbiol. 2016 May 2;82(10):3082-91. doi: 10.1128/AEM.04143-15. Print 2016 May 15. Appl Environ Microbiol. 2016. PMID: 27016564 Free PMC article.
-
Effects of luxCDABEG induction in Vibrio fischeri: enhancement of symbiotic colonization and conditional attenuation of growth in culture.Arch Microbiol. 2008 Aug;190(2):169-83. doi: 10.1007/s00203-008-0387-1. Epub 2008 Jun 3. Arch Microbiol. 2008. PMID: 18521572 Free PMC article.
-
Control of biofilm formation and colonization in Vibrio fischeri: a role for partner switching?Environ Microbiol. 2010 Aug;12(8):2051-9. doi: 10.1111/j.1462-2920.2010.02269.x. Epub 2010 Jun 9. Environ Microbiol. 2010. PMID: 21966901 Free PMC article. Review.
-
The type-VI secretion system of the beneficial symbiont Vibrio fischeri.Microbiology (Reading). 2023 Feb;169(2):001302. doi: 10.1099/mic.0.001302. Microbiology (Reading). 2023. PMID: 36809081 Free PMC article. Review.
Cited by
-
Two-component response regulators of Vibrio fischeri: identification, mutagenesis, and characterization.J Bacteriol. 2007 Aug;189(16):5825-38. doi: 10.1128/JB.00242-07. Epub 2007 Jun 22. J Bacteriol. 2007. PMID: 17586650 Free PMC article.
-
Chromosomally Encoded hok-sok Toxin-Antitoxin System in the Fire Blight Pathogen Erwinia amylovora: Identification and Functional Characterization.Appl Environ Microbiol. 2019 Jul 18;85(15):e00724-19. doi: 10.1128/AEM.00724-19. Print 2019 Aug 1. Appl Environ Microbiol. 2019. PMID: 31101613 Free PMC article.
-
Mutations in ampG and lytic transglycosylase genes affect the net release of peptidoglycan monomers from Vibrio fischeri.J Bacteriol. 2009 Apr;191(7):2012-22. doi: 10.1128/JB.01547-08. Epub 2008 Dec 12. J Bacteriol. 2009. PMID: 19074387 Free PMC article.
-
Relative contributions of norspermidine synthesis and signaling pathways to the regulation of Vibrio cholerae biofilm formation.PLoS One. 2017 Oct 18;12(10):e0186291. doi: 10.1371/journal.pone.0186291. eCollection 2017. PLoS One. 2017. PMID: 29045455 Free PMC article.
-
Cyclic Di-GMP and VpsR Induce the Expression of Type II Secretion in Vibrio cholerae.J Bacteriol. 2017 Sep 5;199(19):e00106-17. doi: 10.1128/JB.00106-17. Print 2017 Oct 1. J Bacteriol. 2017. PMID: 28674069 Free PMC article.
References
-
- Bevis, B. J., and B. S. Glick. 2002. Rapidly maturing variants of the Discosoma red fluorescent protein (DsRed). Nat. Biotechnol. 20:83-87. - PubMed
-
- Boettcher, K. J., and E. G. Ruby. 1994. Occurrence of plasmid DNA in the sepiolid squid symbiont Vibrio fischeri. Curr. Microbiol. 29:279-286.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

