Quantitative PET imaging of tumor integrin alphavbeta3 expression with 18F-FRGD2
- PMID: 16391195
- PMCID: PMC4160026
Quantitative PET imaging of tumor integrin alphavbeta3 expression with 18F-FRGD2
Abstract
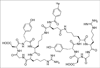
The development of noninvasive methods to visualize and quantify integrin alpha(v)beta(3) expression in vivo appears to be crucial for the success of antiangiogenic therapy based on integrin antagonism. Precise documentation of integrin receptor levels will allow appropriate selection of patients who will most likely benefit from an antiintegrin treatment regimen. Imaging can also be used to provide an optimal dosage and time course for treatment based on receptor occupancy studies. In addition, imaging integrin expression will be important to evaluate antiintegrin treatment efficacy and to develop new therapeutic drugs with favorable tumor targeting and in vivo kinetics. We labeled the dimeric RGD peptide E[c(RGDyK)](2) with (18)F and evaluated its tumor-targeting efficacy and pharmacokinetics of (18)F-FB-E[c(RGDyK)](2) ((18)F-FRGD2).
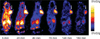
Methods: E[c(RGDyK)](2) was labeled with (18)F by conjugation coupling with N-succinimidyl-4-(18)F-fluorobenzoate ((18)F-SFB) under a slightly basic condition. The in vivo metabolic stability of (18)F-FRGD2 was determined. The diagnostic value after injection of (18)F-FRGD2 was evaluated in various xenograft models by dynamic microPET followed by ex vivo quantification of tumor integrin level.
Results: Starting with (18)F(-) Kryptofix 2.2.2./K(2)CO(3) solution, the total reaction time for (18)F-FRGD2, including final high-performance liquid chromatography purification, is about 200 +/- 20 min. Typical decay-corrected radiochemical yield is 23% +/- 2% (n = 20). (18)F-FRGD2 is metabolically stable. The binding potential extrapolated from graphical analysis of PET data and Logan plot correlates well with the receptor density measured by sodium dodecyl sulfate polyacrylamide electrophoresis and autoradiography in various xenograft models. The tumor-to-background ratio at 1 h after injection of (18)F-FRGD2 also gives a good linear relationship with the tumor tissue integrin level.
Conclusion: The dimeric RGD peptide tracer (18)F-FRGD2, with high integrin specificity and favorable excretion profile, may be translated into the clinic for imaging integrin alpha(v)beta(3) expression. The binding potential calculated from simplified tracer kinetic modeling such as the Logan plot appears to be an excellent indicator of tumor integrin density.
Figures




References
-
- Danen EH. Integrins: regulators of tissue function and cancer progression. Curr Pharm Des. 2005;11:881–891. - PubMed
-
- Hynes RO. Integrins: bidirectional, allosteric signaling machines. Cell. 2002;110:673–687. - PubMed
-
- Robinson SD, Reynolds LE, Wyder L, et al. b3-Integrin regulates vascular endothelial growth factor-A-dependent permeability. Arterioscler Thromb Vasc Biol. 2004;24:2108–2114. - PubMed
-
- Friedlander M, Brooks PC, Shaffer RW, et al. Definition of two angiogenic pathways by distinct αv integrins. Science. 1995;270:1500–1502. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
