Early preplasma cells define a tolerance checkpoint for autoreactive B cells
- PMID: 16393962
- PMCID: PMC3709567
- DOI: 10.4049/jimmunol.176.2.790
Early preplasma cells define a tolerance checkpoint for autoreactive B cells
Abstract
Ab-secreting plasma cells (PCs) are the effectors of humoral immunity. In this study, we describe regulation of autoreactive B cells specific for the ribonucleoprotein Smith (Sm) at an early pre-PC stage. These cells are defined by the expression of the PC marker CD138 and normal levels of CD19 and B220. They are present at a high frequency in normal mouse spleen and bone marrow, are Ag dependent, and are located predominantly along the T cell-B cell border and near bridging channels. Anti-Sm pre-PCs also occur at a high frequency in nonautoimmune mice and show additional phenotypic characteristics of PC differentiation. However, while some of these pre-PCs are Ab-secreting cells, those specific for Sm are not, indicating regulation. Consistent with this, anti-Sm pre-PCs have a higher turnover rate and higher frequency of cell death than those that do not bind Sm. Regulation of anti-Sm pre-PCs occurs upstream of the transcriptional repressor, B lymphocyte-induced maturation protein-1, expression. Regulation at this stage is overcome in autoimmune MRL/lpr mice and is accompanied by an altered B lymphocyte stimulator receptor profile. These data reveal a new B cell tolerance checkpoint that is overcome in autoimmunity.
Figures

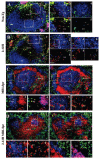


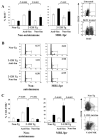
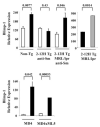
 ) B cells in 2-12H MRL/lpr. In the lower graph are relative Blimp-1 levels from sorted CD138− and CD138int B cells from MD4 and MD4 × ML5 mice. Data are representative of two independent experiments using pooled mice. Values of p are shown for the indicated comparisons.
) B cells in 2-12H MRL/lpr. In the lower graph are relative Blimp-1 levels from sorted CD138− and CD138int B cells from MD4 and MD4 × ML5 mice. Data are representative of two independent experiments using pooled mice. Values of p are shown for the indicated comparisons.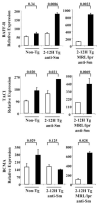
Similar articles
-
Anti-Sm B cell differentiation in Ig transgenic MRL/Mp-lpr/lpr mice: altered differentiation and an accelerated response.J Immunol. 2001 Apr 15;166(8):5292-9. doi: 10.4049/jimmunol.166.8.5292. J Immunol. 2001. PMID: 11290816
-
Lupus-specific antiribonucleoprotein B cell tolerance in nonautoimmune mice is maintained by differentiation to B-1 and governed by B cell receptor signaling thresholds.J Immunol. 2001 Feb 15;166(4):2412-9. doi: 10.4049/jimmunol.166.4.2412. J Immunol. 2001. PMID: 11160300
-
Differential involvement of the transcription factor Blimp-1 in T cell-independent and -dependent B cell differentiation to plasma cells.J Immunol. 1999 Jul 15;163(2):611-7. J Immunol. 1999. PMID: 10395648
-
Determinations of B cell fate in immunity and autoimmunity.Curr Dir Autoimmun. 2005;8:1-24. doi: 10.1159/000082084. Curr Dir Autoimmun. 2005. PMID: 15564715 Review.
-
Regulation of B-cell commitment to plasma cells or to memory B cells.Semin Immunol. 1997 Aug;9(4):235-40. doi: 10.1006/smim.1997.0080. Semin Immunol. 1997. PMID: 9237929 Review.
Cited by
-
Somatic hypermutation as a generator of antinuclear antibodies in a murine model of systemic autoimmunity.J Exp Med. 2010 Sep 27;207(10):2225-37. doi: 10.1084/jem.20092712. Epub 2010 Aug 30. J Exp Med. 2010. PMID: 20805563 Free PMC article.
-
Activation-induced deaminase heterozygous MRL/lpr mice are delayed in the production of high-affinity pathogenic antibodies and in the development of lupus nephritis.Immunology. 2009 Jan;126(1):102-13. doi: 10.1111/j.1365-2567.2008.02882.x. Epub 2008 Jun 20. Immunology. 2009. PMID: 18624728 Free PMC article.
-
Low-Level Expression of CD138 Marks Naturally Arising Anergic B Cells.Immune Netw. 2022 Oct 24;22(6):e50. doi: 10.4110/in.2022.22.e50. eCollection 2022 Dec. Immune Netw. 2022. PMID: 36627940 Free PMC article.
-
B cell receptor revision diminishes the autoreactive B cell response after antigen activation in mice.J Clin Invest. 2008 Aug;118(8):2896-907. doi: 10.1172/JCI35618. J Clin Invest. 2008. PMID: 18636122 Free PMC article.
-
The role of B cells in lupus pathogenesis.Int J Biochem Cell Biol. 2010 Apr;42(4):543-50. doi: 10.1016/j.biocel.2009.10.011. Epub 2009 Oct 20. Int J Biochem Cell Biol. 2010. PMID: 19850148 Free PMC article. Review.
References
-
- Calame KL, Lin KI, Tunyaplin C. Regulatory mechanisms that determine the development and function of plasma cells. Annu. Rev. Immunol. 2003;21:205–230. - PubMed
-
- MacLennan IC, Toellner KM, Cunningham AF, Serre K, Sze DM, Zuniga E, Cook MC, Vinuesa CG. Extrafollicular antibody responses. Immunol. Rev. 2003;194:8–18. - PubMed
-
- Tarte K, Zhan F, De Vos J, Klein B, Shaughnessy J., Jr. Gene expression profiling of plasma cells and plasmablasts: toward a better understanding of the late stages of B-cell differentiation. Blood. 2003;102:592–600. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

