PGAP2 is essential for correct processing and stable expression of GPI-anchored proteins
- PMID: 16407401
- PMCID: PMC1382328
- DOI: 10.1091/mbc.e05-11-1005
PGAP2 is essential for correct processing and stable expression of GPI-anchored proteins
Abstract
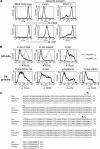
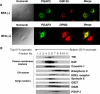
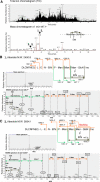
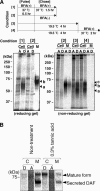
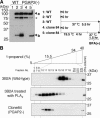
Biosynthesis of glycosylphosphatidylinositol-anchored proteins (GPI-APs) in the ER has been extensively studied, whereas the molecular events during the transport of GPI-APs from the ER to the cell surface are poorly understood. Here, we established new mutant cell lines whose surface expressions of GPI-APs were greatly decreased despite normal biosynthesis of GPI-APs in the ER. We identified a gene responsible for this defect, designated PGAP2 (for Post-GPI-Attachment to Proteins 2), which encoded a Golgi/ER-resident membrane protein. The low surface expression of GPI-APs was due to their secretion into the culture medium. GPI-APs were modified/cleaved by two reaction steps in the mutant cells. First, the GPI anchor was converted to lyso-GPI before exiting the trans-Golgi network. Second, lyso-GPI-APs were cleaved by a phospholipase D after transport to the plasma membrane. Therefore, PGAP2 deficiency caused transport to the cell surface of lyso-GPI-APs that were sensitive to a phospholipase D. These results demonstrate that PGAP2 is involved in the processing of GPI-APs required for their stable expression at the cell surface.
Figures

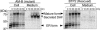
References
-
- Ahmed, S. N., Brown, D. A., and London, E. (1997). On the origin of sphingolipid/cholesterol-rich detergent-insoluble cell membranes: physiological concentrations of cholesterol and sphingolipid induce formation of a detergent-insoluble, liquid-ordered lipid phase in model membranes. Biochemistry 36, 10944–10953. - PubMed
-
- Brewis, I. A., Ferguson, M. A., Mehlert, A., Turner, A. J., and Hooper, N. M. (1995). Structures of the glycosyl-phosphatidylinositol anchors of porcine and human renal membrane dipeptidase. Comprehensive structural studies on the porcine anchor and interspecies comparison of the glycan core structures. J. Biol. Chem. 270, 22946–22956. - PubMed
-
- Brown, D. A., and Rose, J. K. (1992). Sorting of GPI-anchored proteins to glycolipid-enriched membrane subdomains during transport to the apical cell surface. Cell 68, 533–544. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

