Different effects on fast exocytosis induced by synaptotagmin 1 and 2 isoforms and abundance but not by phosphorylation
- PMID: 16407561
- PMCID: PMC6674391
- DOI: 10.1523/JNEUROSCI.2589-05.2006
Different effects on fast exocytosis induced by synaptotagmin 1 and 2 isoforms and abundance but not by phosphorylation
Abstract
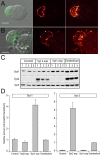
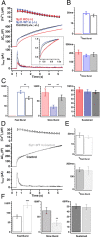
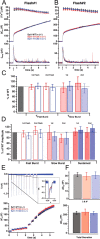
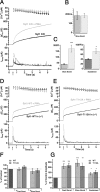
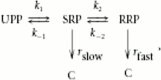
Synaptotagmins comprise a large protein family, of which synaptotagmin 1 (Syt1) is a Ca2+ sensor for fast exocytosis, and its close relative, synaptotagmin 2 (Syt2), is assumed to serve similar functions. Chromaffin cells express Syt1 but not Syt2. We compared secretion from chromaffin cells from Syt1 null mice overexpressing either Syt isoform. High time-resolution capacitance measurement showed that Syt1 null cells lack the exocytotic phase corresponding to the readily-releasable pool (RRP) of vesicles. Comparison with the amperometric signal confirmed that the missing phase of exocytosis consists of catecholamine-containing vesicles. Overexpression of Syt1 rescued the RRP and increased its size above wild-type values, whereas the size of the slowly releasable pool decreased, indicating that the availability of Syt1 regulates the relative size of the two releasable pools. The RRP was also rescued by Syt2 overexpression, but the kinetics of fusion was slightly slower than in cells expressing Syt1. Biochemical experiments showed that Syt2 has a slightly lower Ca2+ affinity for phospholipid binding than Syt1 because of a difference in the C2A domain. These data constitute evidence for the function of Syt1 and Syt2 as alternative, but not identical, calcium-sensors for RRP fusion. By overexpression of Syt1 mutated in the shared PKC/calcium/calmodulin-dependent kinase phosphorylation site, we show that phorbol esters act independently and upstream of Syt1 to regulate the size of the releasable pools. We conclude that exocytosis from mouse chromaffin cells can be modified by the differential expression of Syt isoforms and by Syt abundance but not by phosphorylation of Syt1.
Figures



Similar articles
-
Control of exocytosis by synaptotagmins and otoferlin in auditory hair cells.J Neurosci. 2010 Oct 6;30(40):13281-90. doi: 10.1523/JNEUROSCI.2528-10.2010. J Neurosci. 2010. PMID: 20926654 Free PMC article.
-
A Post-Docking Role of Synaptotagmin 1-C2B Domain Bottom Residues R398/399 in Mouse Chromaffin Cells.J Neurosci. 2015 Oct 21;35(42):14172-82. doi: 10.1523/JNEUROSCI.1911-15.2015. J Neurosci. 2015. PMID: 26490858 Free PMC article.
-
Phosphomimetic mutation of Ser-187 of SNAP-25 increases both syntaxin binding and highly Ca2+-sensitive exocytosis.J Gen Physiol. 2007 Mar;129(3):233-44. doi: 10.1085/jgp.200609685. J Gen Physiol. 2007. PMID: 17325194 Free PMC article.
-
Synaptotagmin regulates mast cell functions.Immunol Rev. 2001 Feb;179:25-34. doi: 10.1034/j.1600-065x.2001.790103.x. Immunol Rev. 2001. PMID: 11292024 Review.
-
Exocytosis and synaptic vesicle function.Compr Physiol. 2014 Jan;4(1):149-75. doi: 10.1002/cphy.c130021. Compr Physiol. 2014. PMID: 24692137 Review.
Cited by
-
Complexin facilitates exocytosis and synchronizes vesicle release in two secretory model systems.J Physiol. 2013 May 15;591(10):2463-73. doi: 10.1113/jphysiol.2012.244517. Epub 2013 Feb 11. J Physiol. 2013. PMID: 23401610 Free PMC article.
-
Synaptotagmin-2 is a reliable marker for parvalbumin positive inhibitory boutons in the mouse visual cortex.PLoS One. 2012;7(4):e35323. doi: 10.1371/journal.pone.0035323. Epub 2012 Apr 23. PLoS One. 2012. PMID: 22539967 Free PMC article.
-
Genetic analysis of synaptotagmin 2 in spontaneous and Ca2+-triggered neurotransmitter release.EMBO J. 2006 May 17;25(10):2039-50. doi: 10.1038/sj.emboj.7601103. Epub 2006 Apr 27. EMBO J. 2006. PMID: 16642042 Free PMC article.
-
Vesicle pools: lessons from adrenal chromaffin cells.Front Synaptic Neurosci. 2011 Feb 1;3:2. doi: 10.3389/fnsyn.2011.00002. eCollection 2011. Front Synaptic Neurosci. 2011. PMID: 21423410 Free PMC article.
-
Brain Acetyl-CoA Production and Phosphorylation of Cytoskeletal Proteins Are Targets of CYP46A1 Activity Modulation and Altered Sterol Flux.Neurotherapeutics. 2021 Jul;18(3):2040-2060. doi: 10.1007/s13311-021-01079-6. Epub 2021 Jul 7. Neurotherapeutics. 2021. PMID: 34235635 Free PMC article.
References
-
- Albillos A, Dernick G, Horstmann H, Almers W, Alvarez de Toledo G, Lindau M (1997) The exocytotic event in chromaffin cells revealed by patch amperometry. Nature 389: 509-512. - PubMed
-
- Aldea M, Jun K, Shin HS, Andres-Mateos E, Solis-Garrido LM, Montiel C, Garcia AG, Albillos A (2002) A perforated patch-clamp study of calcium currents and exocytosis in chromaffin cells of wild-type and α1A knockout mice. J Neurochem 81: 911-921. - PubMed
-
- Ashery U, Betz A, Xu T, Brose N, Rettig J (1999) An efficient method for infection of adrenal chromaffin cells using the Semliki Forest virus gene expression system. Eur J Cell Biol 78: 525-532. - PubMed
-
- Augustine GJ (2001) How does calcium trigger neurotransmitter release? Curr Opin Neurobiol 11: 320-326. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
