Canine adenovirus vectors for lung-directed gene transfer: efficacy, immune response, and duration of transgene expression using helper-dependent vectors
- PMID: 16415025
- PMCID: PMC1346928
- DOI: 10.1128/JVI.80.3.1487-1496.2006
Canine adenovirus vectors for lung-directed gene transfer: efficacy, immune response, and duration of transgene expression using helper-dependent vectors
Abstract
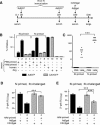
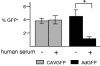
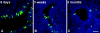
A major hurdle to the successful clinical use of some viral vectors relates to the innate, adaptive, and memory immune responses that limit the efficiency and duration of transgene expression. Some of these drawbacks may be circumvented by using vectors derived from nonhuman viruses such as canine adenovirus type 2 (CAV-2). Here, we evaluated the potential of CAV-2 vectors for gene transfer to the respiratory tract. We found that CAV-2 transduction was efficient in vivo in the mouse respiratory tract, and ex vivo in well-differentiated human pulmonary epithelia. Notably, the in vivo and ex vivo efficiency was poorly inhibited by sera from mice immunized with a human adenovirus type 5 (HAd5, a ubiquitous human pathogen) vector or by human sera containing HAd5 neutralizing antibodies. Following intranasal instillation in mice, CAV-2 vectors also led to a lower level of inflammatory cytokine secretion and cellular infiltration compared to HAd5 vectors. Moreover, CAV-2 transduction efficiency was increased in vitro in human pulmonary cells and in vivo in the mouse respiratory tract by FK228, a histone deacetylase inhibitor. Finally, by using a helper-dependent CAV-2 vector, we increased the in vivo duration of transgene expression to at least 3 months in immunocompetent mice without immunosuppression. Our data suggest that CAV-2 vectors may be efficient and safe tools for long-term clinical gene transfer to the respiratory tract.
Figures

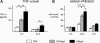
Similar articles
-
The conundrum between immunological memory to adenovirus and their use as vectors in clinical gene therapy.Mol Biotechnol. 2006 Oct;34(2):247-56. doi: 10.1385/MB:34:2:247. Mol Biotechnol. 2006. PMID: 17172670 Review.
-
Circumvention of vector-specific neutralizing antibody response by alternating use of human and non-human adenoviruses: implications in gene therapy.Virology. 2000 Jun 20;272(1):159-67. doi: 10.1006/viro.2000.0350. Virology. 2000. PMID: 10873758
-
Contrasting effects of human, canine, and hybrid adenovirus vectors on the phenotypical and functional maturation of human dendritic cells: implications for clinical efficacy.J Virol. 2007 Apr;81(7):3272-84. doi: 10.1128/JVI.01530-06. Epub 2007 Jan 17. J Virol. 2007. PMID: 17229706 Free PMC article.
-
SGSH gene transfer in mucopolysaccharidosis type IIIA mice using canine adenovirus vectors.Mol Genet Metab. 2010 Jun;100(2):168-75. doi: 10.1016/j.ymgme.2010.02.006. Epub 2010 Feb 14. Mol Genet Metab. 2010. PMID: 20231109
-
CAV-2--why a canine virus is a neurobiologist's best friend.Curr Opin Pharmacol. 2015 Oct;24:86-93. doi: 10.1016/j.coph.2015.08.004. Epub 2015 Aug 25. Curr Opin Pharmacol. 2015. PMID: 26298516 Review.
Cited by
-
Lentivirus vector can be readministered to nasal epithelia without blocking immune responses.J Virol. 2008 Nov;82(21):10684-92. doi: 10.1128/JVI.00227-08. Epub 2008 Sep 3. J Virol. 2008. PMID: 18768988 Free PMC article.
-
Canine recombinant adenovirus vector induces an immunogenicity-related gene expression profile in skin-migrated CD11b⁺ -type DCs.PLoS One. 2012;7(12):e52513. doi: 10.1371/journal.pone.0052513. Epub 2012 Dec 26. PLoS One. 2012. PMID: 23300693 Free PMC article.
-
Central nervous system delivery of helper-dependent canine adenovirus corrects neuropathology and behavior in mucopolysaccharidosis type VII mice.Hum Gene Ther. 2014 Mar;25(3):199-211. doi: 10.1089/hum.2013.152. Epub 2014 Jan 23. Hum Gene Ther. 2014. PMID: 24299455 Free PMC article.
-
Recent advances in gene therapy for lysosomal storage disorders.Appl Clin Genet. 2015 Jun 24;8:157-69. doi: 10.2147/TACG.S57682. eCollection 2015. Appl Clin Genet. 2015. PMID: 26170711 Free PMC article. Review.
-
Sensory cortical control of movement.Nat Neurosci. 2020 Jan;23(1):75-84. doi: 10.1038/s41593-019-0536-7. Epub 2019 Nov 18. Nat Neurosci. 2020. PMID: 31740813
References
-
- Amalfitano, A., and R. J. Parks. 2002. Separating fact from fiction: assessing the potential of modified adenovirus vectors for use in human gene therapy. Curr. Gene Ther. 2:111-133. - PubMed
-
- Amin, R., R. Wilmott, Y. Schwarz, B. Trapnell, and J. Stark. 1995. Replication-deficient adenovirus induces expression of interleukin-8 by airway epithelial cells in vitro. Hum. Gene Ther. 6:145-153. - PubMed
-
- Ayers, M. M., and P. K. Jeffery. 1988. Proliferation and differentiation in mammalian airway epithelium. Eur. Respir. J. 1:58-80. - PubMed
-
- Bergelson, J. M., J. A. Cunningham, G. Droguett, E. A. Kurt-Jones, A. Krithivas, J. S. Hong, M. S. Horwitz, R. L. Crowell, and R. W. Finberg. 1997. Isolation of a common receptor for Coxsackie B viruses and adenoviruses 2 and 5. Science 275:1320-1323. - PubMed
-
- Bowen, G. P., S. L. Borgland, M. Lam, T. A. Libermann, N. C. Wong, and D. A. Muruve. 2002. Adenovirus vector-induced inflammation: capsid-dependent induction of the C-C chemokine RANTES requires NF-kappa B. Hum. Gene Ther. 13:367-379. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

