Direct stimulation of translation by the multifunctional herpesvirus ICP27 protein
- PMID: 16415034
- PMCID: PMC1346932
- DOI: 10.1128/JVI.80.3.1588-1591.2006
Direct stimulation of translation by the multifunctional herpesvirus ICP27 protein
Abstract
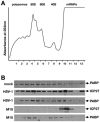
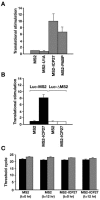
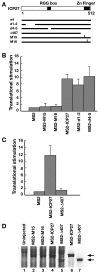
Herpes simplex virus type 1 (HSV-1) ICP27 protein is an essential regulator of viral gene expression with roles at various levels of RNA metabolism in the nucleus. Using the tethered function assay, we showed a cytoplasmic activity for ICP27 in directly enhancing mRNA translation in vivo in the absence of other viral factors. The region of ICP27 required for translational stimulation maps to the C terminus. Furthermore, in infected cells, ICP27 is associated with polyribosomes, indicating a function in translation during the lytic cycle.
Figures
Similar articles
-
Control of VP16 translation by the herpes simplex virus type 1 immediate-early protein ICP27.J Virol. 2005 Apr;79(7):4120-31. doi: 10.1128/JVI.79.7.4120-4131.2005. J Virol. 2005. PMID: 15767413 Free PMC article.
-
Processing of alpha-globin and ICP0 mRNA in cells infected with herpes simplex virus type 1 ICP27 mutants.J Virol. 2000 Aug;74(16):7307-19. doi: 10.1128/jvi.74.16.7307-7319.2000. J Virol. 2000. PMID: 10906184 Free PMC article.
-
ICP27 phosphorylation site mutants display altered functional interactions with cellular export factors Aly/REF and TAP/NXF1 but are able to bind herpes simplex virus 1 RNA.J Virol. 2010 Mar;84(5):2212-22. doi: 10.1128/JVI.01388-09. Epub 2009 Dec 16. J Virol. 2010. PMID: 20015986 Free PMC article.
-
The many roles of the regulatory protein ICP27 during herpes simplex virus infection.Front Biosci. 2008 May 1;13:5241-56. doi: 10.2741/3078. Front Biosci. 2008. PMID: 18508584 Review.
-
Role of human herpesvirus homologs of infected cell protein 27 (ICP27) in the biogenesis, processing, and maturation of mRNAs.mBio. 2025 Apr 9;16(4):e0029125. doi: 10.1128/mbio.00291-25. Epub 2025 Mar 4. mBio. 2025. PMID: 40035535 Free PMC article. Review.
Cited by
-
When Poly(A) Binding Proteins Meet Viral Infections, Including SARS-CoV-2.J Virol. 2022 Apr 13;96(7):e0013622. doi: 10.1128/jvi.00136-22. Epub 2022 Mar 16. J Virol. 2022. PMID: 35293770 Free PMC article. Review.
-
Preventing translational inhibition from ribosomal protein insufficiency by a herpes simplex virus-encoded ribosome-associated protein.Proc Natl Acad Sci U S A. 2021 Nov 9;118(45):e2025546118. doi: 10.1073/pnas.2025546118. Proc Natl Acad Sci U S A. 2021. PMID: 34725147 Free PMC article.
-
Poly(A)-binding protein 1 partially relocalizes to the nucleus during herpes simplex virus type 1 infection in an ICP27-independent manner and does not inhibit virus replication.J Virol. 2010 Sep;84(17):8539-48. doi: 10.1128/JVI.00668-10. Epub 2010 Jun 23. J Virol. 2010. PMID: 20573819 Free PMC article.
-
Human cytomegalovirus UL84 protein contains two nuclear export signals and shuttles between the nucleus and the cytoplasm.J Virol. 2006 Oct;80(20):10274-80. doi: 10.1128/JVI.00995-06. J Virol. 2006. PMID: 17005707 Free PMC article.
-
Kaposi's sarcoma-associated herpesvirus ORF57 protein interacts with PYM to enhance translation of viral intronless mRNAs.EMBO J. 2010 Jun 2;29(11):1851-64. doi: 10.1038/emboj.2010.77. Epub 2010 Apr 30. EMBO J. 2010. PMID: 20436455 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

