OX40 interactions in gastrointestinal nematode infection
- PMID: 16423046
- PMCID: PMC1782197
- DOI: 10.1111/j.1365-2567.2005.02271.x
OX40 interactions in gastrointestinal nematode infection
Abstract
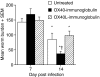
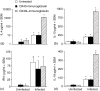
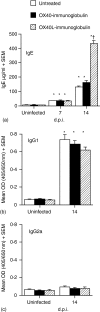
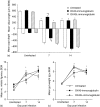
The immune expulsion of gastrointestinal nematode parasites is usually associated with T helper type 2 (Th2) responses, but the effector mechanisms directly responsible for parasite loss have not been elucidated. The intestinal inflammatory response accompanying infection with gastrointestinal helminths is thought to be a contributory factor leading to the expulsion of the parasite. However, we have shown that the intestinal inflammation, which is controlled by interleukin (IL)-4, is not required for parasite expulsion. OX40-OX40 ligand (L) signals have been shown to be important for the development of Th2 immune responses but are also involved in a number of inflammatory diseases including those of the intestine. Here, we have investigated the effect of OX40 and OX40L fusion protein treatment on the induction of protective Th2 responses and enteropathy following infection with the gastrointestinal nematode Trichinella spiralis. Treatment with an OX40-immunoglobulin (Ig) blocking fusion protein resulted in enhanced expulsion of the parasite and an increase in the accompanying mastocytosis, despite unaltered levels of Th2 cytokines. Furthermore, there was a delay in the villus atrophy and crypt hyperplasia usually associated with this infection. In contrast, levels of Th2 cytokines were greatly up-regulated in mice treated with an OX40L-Ig activating fusion protein, yet the expulsion of the parasite and the enteropathy were unaffected. Therefore, OX40 ligation potentiates the Th2 response without enhancing host protective immune responses, whereas blocking the OX40-OX40L interaction enhances host protection without promoting Th2 cytokine responses during Trichinella spiralis infection.
Figures
Similar articles
-
Nitric oxide mediates intestinal pathology but not immune expulsion during Trichinella spiralis infection in mice.J Immunol. 2000 Apr 15;164(8):4229-34. doi: 10.4049/jimmunol.164.8.4229. J Immunol. 2000. PMID: 10754319
-
The role of OX40 ligand interactions in the development of the Th2 response to the gastrointestinal nematode parasite Heligmosomoides polygyrus.J Immunol. 2003 Jan 1;170(1):384-93. doi: 10.4049/jimmunol.170.1.384. J Immunol. 2003. PMID: 12496423
-
The role of IL-4, IL-13 and IL-4Ralpha in the development of protective and pathological responses to Trichinella spiralis.Parasite Immunol. 2007 Feb;29(2):81-91. doi: 10.1111/j.1365-3024.2006.00920.x. Parasite Immunol. 2007. PMID: 17241396
-
Immunopathology of intestinal helminth infection.Parasite Immunol. 2000 Dec;22(12):605-12. doi: 10.1046/j.1365-3024.2000.00344.x. Parasite Immunol. 2000. PMID: 11123752 Review.
-
[Effect of ketotifen on the immune response in BALB/c mice infected with Trichinella spiralis].Wiad Parazytol. 2000;46(2):217-24. Wiad Parazytol. 2000. PMID: 16886340 Review. Polish.
Cited by
-
OX40 Expression in Eosinophils Aggravates OVA-Induced Eosinophilic Gastroenteritis.Front Immunol. 2022 Jun 2;13:841141. doi: 10.3389/fimmu.2022.841141. eCollection 2022. Front Immunol. 2022. PMID: 35720294 Free PMC article.
-
Control of immunity by the TNFR-related molecule OX40 (CD134).Annu Rev Immunol. 2010;28:57-78. doi: 10.1146/annurev-immunol-030409-101243. Annu Rev Immunol. 2010. PMID: 20307208 Free PMC article. Review.
-
Functional investigation of a QTL affecting resistance to Haemonchus contortus in sheep.Vet Res. 2014 Jun 17;45(1):68. doi: 10.1186/1297-9716-45-68. Vet Res. 2014. PMID: 24939584 Free PMC article.
-
OX40L-OX40 Interactions: A Possible Target for Gastrointestinal Autoimmune Diseases.N Am J Med Sci. 2012 Nov;4(11):533-6. doi: 10.4103/1947-2714.103311. N Am J Med Sci. 2012. PMID: 23181223 Free PMC article.
References
-
- Chan MS, Medley GF, Jamison D, Bundy DA. The evaluation of potential global morbidity attributable to intestinal nematode infections. Parasitology. 1994;109:373–87. - PubMed
-
- Manson-Smith DF, Bruce RG, Parrott DM. Villous atrophy and expulsion of intestinal Trichinella spiralis are mediated by T cells. Cell Immunol. 1979;47:285–92. - PubMed
-
- Garside P, Grencis RK, Mowat AM. T lymphocyte dependent enteropathy in murine Trichinella spiralis infection. Parasite Immunol. 1992;14:217–25. - PubMed
-
- Finkelman FD, Shea-Donohue T, Morris SC, Gildea L, Strait R, Madden KB, Schopf L, Urban JF., Jr Interleukin-4- and interleukin-13-mediated host protection against intestinal nematode parasites. Immunol Rev. 2004;201:139–55. - PubMed
-
- Lawrence CE. Is there a common mechanism of gastrointestinal nematode expulsion? Parasite Immunol. 2003;25:271–81. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

