High-dose weekly AmBisome antifungal prophylaxis in pediatric patients undergoing hematopoietic stem cell transplantation: a pharmacokinetic study
- PMID: 16443521
- PMCID: PMC4912056
- DOI: 10.1016/j.bbmt.2005.10.010
High-dose weekly AmBisome antifungal prophylaxis in pediatric patients undergoing hematopoietic stem cell transplantation: a pharmacokinetic study
Abstract
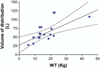
Disseminated fungal infection causes significant morbidity and mortality in children undergoing hematopoietic stem cell transplantation (HSCT). The widespread use of prophylactic oral triazoles has limitations of poor absorption, interindividual variability in metabolism, and hepatic toxicity. AmBisome (amphotericin B liposomal complex) has a better safety profile than the parent drug amphotericin B and produces higher plasma and tissue concentrations. We hypothesized that once-weekly high-dose AmBisome therapy could provide adequate fungal prophylaxis for immunocompromised children undergoing HSCT. We performed a pharmacokinetic pilot study to determine whether once-weekly high-dose AmBisome administration would result in effective concentrations throughout the dosing interval. A total of 14 children (median age, 3 years, 1 month; range, 4.5 months-9 years, 9 months) undergoing HSCT received once-weekly intravenous AmBisome prophylaxis (10 mg/kg as a 2-hour infusion). Blood samples for pharmacokinetic measurements were drawn around the first and the fourth weekly doses. The concentration of non-lipid-complexed amphotericin in plasma was determined by a validated bioassay. Pharmacokinetic parameters after single doses and during steady state were calculated using standard noncompartmental methods. AmBisome was well tolerated at this dose. Complete pharmacokinetic profiles for weeks 1 and 4 were obtained in 12 patients. The half-life calculated in this pediatric population was shorter on average than reported in adults (45 hours vs 152 hours). The volume of distribution correlated best with body weight (R(2) = .55), and clearance was best predicted by initial serum creatinine level (R(2) = .19). Mean (+/- standard deviation) individual plasma trough concentrations were 0.23 (0.13) mg/L after single doses and 0.47 (0.41) mg/L after multiple doses. Mean steady-state area under the curve was higher at week 4 than after a single dose (P < .05). Single-dose and steady-state pharmacokinetic profiles were similar in 8 patients, whereas in 4 patients the week 4 profile showed nonlinear elimination. However, plasma concentrations at 7 days (Cmin) were not significantly different after the first and fourth doses, suggesting no significant accumulation over the course of therapy. Our data show measurable amphotericin B plasma concentrations 7 days after high-dose infusion of AmBisome. This suggests that once-weekly dosing, as described in this study, may provide useful protection against fungal infections.
Figures


Similar articles
-
Alternate-day micafungin antifungal prophylaxis in pediatric patients undergoing hematopoietic stem cell transplantation: a pharmacokinetic study.Biol Blood Marrow Transplant. 2010 Oct;16(10):1458-62. doi: 10.1016/j.bbmt.2010.05.002. Epub 2010 Jun 20. Biol Blood Marrow Transplant. 2010. PMID: 20546908 Clinical Trial.
-
Liposomal amphotericin B (AmBisome) for fungal infections in immunocompromised adults and children.Clin Microbiol Infect. 2001;7 Suppl 2:68-79. doi: 10.1111/j.1469-0691.2001.tb00012.x. Clin Microbiol Infect. 2001. PMID: 11525221 Review.
-
Micafungin antifungal prophylaxis in children undergoing HSCT: can we give higher doses, less frequently? A pharmacokinetic study.J Antimicrob Chemother. 2018 Jun 1;73(6):1651-1658. doi: 10.1093/jac/dky030. J Antimicrob Chemother. 2018. PMID: 29481593
-
Tolerability and outcome of once weekly liposomal amphotericin B for the prevention of invasive fungal infections in hematopoietic stem cell transplant patients with graft-versus-host disease.J Oncol Pharm Pract. 2016 Apr;22(2):228-34. doi: 10.1177/1078155214560920. Epub 2014 Dec 3. J Oncol Pharm Pract. 2016. PMID: 25471252 Free PMC article.
-
AmBisome (liposomal amphotericin B): a comparative review.J Clin Pharmacol. 1998 Jul;38(7):583-92. doi: 10.1002/j.1552-4604.1998.tb04464.x. J Clin Pharmacol. 1998. PMID: 9702842 Review.
Cited by
-
Prospective phase II single-center study of the safety of a single very high dose of liposomal amphotericin B for antifungal prophylaxis in patients with acute myeloid leukemia.Antimicrob Agents Chemother. 2013 Jun;57(6):2596-602. doi: 10.1128/AAC.00155-13. Epub 2013 Mar 25. Antimicrob Agents Chemother. 2013. PMID: 23529741 Free PMC article. Clinical Trial.
-
Caspofungin as antifungal prophylaxis in pediatric patients undergoing allogeneic hematopoietic stem cell transplantation: a retrospective analysis.BMC Infect Dis. 2012 Jul 2;12:151. doi: 10.1186/1471-2334-12-151. BMC Infect Dis. 2012. PMID: 22747637 Free PMC article. Clinical Trial.
-
Cryptococcus neoformans, a global threat to human health.Infect Dis Poverty. 2023 Mar 17;12(1):20. doi: 10.1186/s40249-023-01073-4. Infect Dis Poverty. 2023. PMID: 36932414 Free PMC article. Review.
-
AMBITION-cm: intermittent high dose AmBisome on a high dose fluconazole backbone for cryptococcal meningitis induction therapy in sub-Saharan Africa: study protocol for a randomized controlled trial.Trials. 2015 Jun 17;16:276. doi: 10.1186/s13063-015-0799-6. Trials. 2015. PMID: 26081985 Free PMC article. Clinical Trial.
-
Short-course High-dose Liposomal Amphotericin B for Human Immunodeficiency Virus-associated Cryptococcal Meningitis: A Phase 2 Randomized Controlled Trial.Clin Infect Dis. 2019 Jan 18;68(3):393-401. doi: 10.1093/cid/ciy515. Clin Infect Dis. 2019. PMID: 29945252 Free PMC article. Clinical Trial.
References
-
- Abbasi S, Shenep JL, Hughes WT, et al. Aspergillosis in children with cancer: a 34-year experience. Clin Infect Dis. 1999;29:1210–1219. - PubMed
-
- Groll AH, Müller FM, Piscitelli SC, et al. Lipid formulations of amphotericin B: clinical perspectives for the management of invasive fungal infections in children with cancer. Klin Pädiatr. 1998;210:264–273. - PubMed
-
- Ringden O, Andstrom EE, Remberger M, et al. Prophylaxis and therapy using liposomal amphotericin B (AmBisome) for invasive fungal infections in children undergoing organ or allogeneic bone-marrow transplantation. Pediatr Transplant. 1997;1:124–129. - PubMed
-
- Hovi L, Saarinen-Pihkala UM, Vettenranta K, et al. Invasive fungal infections in pediatric bone marrow transplant recipients: single-center experience of 10 years. Bone Marrow Transplant. 2000;26:999–1004. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

