Virally-directed fluorescent imaging (VFI) can facilitate endoscopic staging
- PMID: 16446989
- PMCID: PMC1435379
- DOI: 10.1007/s00464-005-0259-6
Virally-directed fluorescent imaging (VFI) can facilitate endoscopic staging
Abstract
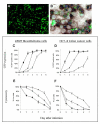
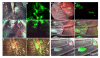
Background: Replication-competent, tumor specific herpes simplex virus NV1066 expresses green fluorescent protein (GFP) in infected cancer cells. We sought to determine the feasibility of GFP-guided imaging technology in the intraoperative detection of small tumor nodules.
Methods: Human cancer cell lines were infected with NV1066 at multiplicities of infection of 0.01, 0.1 and 1. Cancer cell specific infectivity, vector spread and GFP signal intensity were measured by flow cytometry and time-lapse digital imaging (in vitro); and by use of a stereomicroscope and endoscope equipped with a fluorescent filter (in vivo).
Results: NV1066 infected all cancer cell lines and expressed GFP at all MOIs. GFP signal was significantly higher than the autofluorescence of normal cells. One single dose of NV1066 spread within and across body cavities and selectively infected tumor nodules sparing normal tissue. Tumor nodules undetectable by conventional thoracoscopy and laparoscopy were identified by GFP fluorescence.
Conclusion: Virally-directed fluorescent imaging (VFI) is a real-time novel molecular imaging technology that has the potential to enhance the intraoperative detection of endoluminal or endocavitary tumor nodules.
Figures


Similar articles
-
Real-time fluorescence imaging of abdominal, pleural, and lymphatic metastases.Methods Mol Biol. 2012;872:141-57. doi: 10.1007/978-1-61779-797-2_10. Methods Mol Biol. 2012. PMID: 22700409
-
Real-time diagnostic imaging of tumors and metastases by use of a replication-competent herpes vector to facilitate minimally invasive oncological surgery.FASEB J. 2006 Apr;20(6):726-8. doi: 10.1096/fj.05-5316fje. Epub 2006 Feb 8. FASEB J. 2006. PMID: 16467372 Free PMC article.
-
The replication-competent oncolytic herpes simplex mutant virus NV1066 is effective in the treatment of esophageal cancer.Surgery. 2003 Aug;134(2):357-64. doi: 10.1067/msy.2003.244. Surgery. 2003. PMID: 12947341
-
Turning killer into cure -- the story of oncolytic herpes simplex viruses.Discov Med. 2015 Nov;20(111):303-9. Discov Med. 2015. PMID: 26645902 Review.
-
The multiple uses of fluorescent proteins to visualize cancer in vivo.Nat Rev Cancer. 2005 Oct;5(10):796-806. doi: 10.1038/nrc1717. Nat Rev Cancer. 2005. PMID: 16195751 Review.
Cited by
-
Oncolytic Herpes Simplex Virus Prevents Premalignant Lesions from Progressing to Cancer.Mol Ther Oncolytics. 2019 Dec 6;16:1-6. doi: 10.1016/j.omto.2019.11.003. eCollection 2020 Mar 27. Mol Ther Oncolytics. 2019. PMID: 31909180 Free PMC article.
-
Herpes simplex virus 1 as an oncolytic viral therapy for refractory cancers.Front Oncol. 2022 Jul 27;12:940019. doi: 10.3389/fonc.2022.940019. eCollection 2022. Front Oncol. 2022. PMID: 35965554 Free PMC article. Review.
-
Fluorescence-assisted cytological testing (FACT): Ex Vivo viral method for enhancing detection of rare cancer cells in body fluids.Mol Med. 2011;17(7-8):628-34. doi: 10.2119/molmed.2011.00078. Epub 2011 Apr 8. Mol Med. 2011. PMID: 21487639 Free PMC article.
References
-
- Paraskeva PA, Purkayastha S, Darzi A. Laparoscopy for malignancy: current status. Seminars in Laparoscopic Surgery. 2004;11(1):27–36. - PubMed
-
- Parra JL, Reddy KR. Diagnostic laparoscopy. Endoscopy. 2004;36(4):289–93. - PubMed
-
- Corvera CU, Weber SM, Jarnagin WR. Role of laparoscopy in the evaluation of biliary tract cancer. Surgical Oncology Clinics of North America. 2002;11(4):877–91. - PubMed
-
- Asao T, Kuwano H, Mochiki E. Laparoscopic surgery update for gastrointestinal malignancy. Journal of Gastroenterology. 2004;39(4):309–18. - PubMed
-
- Saltzman JR. Section III: endoscopic and other staging techniques. Seminars in Thoracic & Cardiovascular Surgery. 2003;15(2):180–6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

