Reasons for the increase in emerging and re-emerging viral infectious diseases
- PMID: 16448839
- PMCID: PMC7110580
- DOI: 10.1016/j.micinf.2005.06.032
Reasons for the increase in emerging and re-emerging viral infectious diseases
Abstract
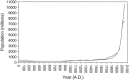
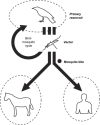
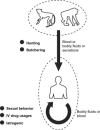
In the past two decades, humans have faced many new viral infectious agents in emerging and re-emerging infectious diseases (EIDs). Many factors contribute to the appearance of EIDs. These factors are complex but can be classified into three different categories: virus factors, human factors, and ecological factors. The factors contributing to the cause of such viral infectious diseases will be systematically reviewed in this article.
Figures

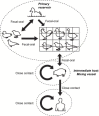
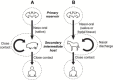
References
-
- Domingo E., Biebrichen C.K., Eigen M., Holland J.J. Landes Bioscience; Georgetown, Texas, USA: 2001. Quasispecies and RNA virus evolution: principles and consequences. <Eurekah.com>.
-
- Mansky L.M. Forward mutation rate of human immunodeficiency virus type 1 in a T-lymphoid cell line. AIDS Res. Hum. Retroviruses. 1996;12:307–314. - PubMed
-
- Consortium TCSme: Molecular evolution of the SARS coronavirus during the course of the SARS epidemic in China. Science. 2004;303:1666–1669. - PubMed
-
- Daar E.S., Richman D.D. Confronting the emergence of drug-resistant HIV type 1: impact of antiretroviral therapy on individual and population resistance. AIDS Res. Hum. Retroviruses. 2005;21:343–357. - PubMed
-
- Hatta M., Gao P., Halfmann P., Kawaoka Y. Molecular basis for high virulence of Hong Kong H5N1 influenza A viruses. Science. 2001;293:1840–1842. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

