Statin wars following coronary revascularization--evidence-based clinical practice?
- PMID: 16450018
- PMCID: PMC2538985
- DOI: 10.1016/s0828-282x(06)70239-2
Statin wars following coronary revascularization--evidence-based clinical practice?
Abstract
Background: Randomized clinical trials (RCTs) have shown that statins provide substantial heath benefits. Pharmaceutical companies spend enormous amounts of money on both clinical trials and marketing. The relative influence of information from clinical trials on physician prescription patterns for statins is unknown.
Objective: To examine the correlation between statin prescription patterns and the quality of evidence from RCTs.
Methods: Using the computerized administrative databases of the Quebec Health Insurance Board, the choice of statin for elderly patients (older than 65 years of age) following a coronary revascularization procedure (percutaneous coronary intervention or coronary artery bypass graft surgery) performed between January 1, 1994, and June 30, 2003, was examined. Prescriptions for each statin were compared with their evidence base obtained from a cumulative systematic literature review of RCTs that recorded mortality as an outcome and were published before December 31, 2002.
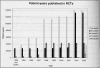
Results: The study cohort comprised 27,979 elderly revascularized patients who received at least one statin prescription. In 1996, the year atorvastatin was introduced, simvastatin and pravastatin had 38.3% and 37.1% of the market share, respectively. By 2003, atorvastatin had 44% of the market share, compared with 29.9% and 24.1% for simvastatin and pravastatin, respectively. In contrast, RCTs published up to the end of 2002 had culminated in 133,341 and 140,565 patient-years of follow-up for simvastatin and pravastatin, respectively, and only 1459 patient-years for atorvastatin.
Conclusions: Prescription patterns regarding the choice of statin do not appear to be determined uniquely from high-quality RCTs. Further research into the other possible determinants of physician prescription patterns is necessary.
CONTEXTE: Les essais cliniques avec hasardisation (ECH) ont montré que les statines avaient un effet salutaire important. Les sociétés pharmaceutiques dépensent des sommes considérables pour les essais cliniques et pour la commercialisation. Toutefois, on ne sait pas dans quelle mesure l’information provenant des essais cliniques influence les médecins dans leurs habitudes de prescription des statines.
BUT: Étudier la corrélation entre les prescriptions de statines et la qualité des données provenant des ECH.
MÉTHODE: Nous avons examiné, à partir de la base de données administratives de la Régie de l’assurance maladie du Québec, le type de statines prescrit à des patients âgés (plus de 65 ans), après une revascularisation coronarienne (intervention coronarienne percutanée ou pontage coronarien) pratiquée entre le 1er janvier 1994 et le 30 juin 2003. Les prescriptions pour chacune des statines ont été comparées aux données obtenues par un examen systématique de l’ensemble de la documentation sur les ECH qui avaient pour critère la mortalité et qui avaient été publiés avant le 31 décembre 2002.
RÉSULTATS: La cohorte à l’étude comptait 27 979 patients âgés qui avaient subi une revascularisation et qui avaient reçu au moins une ordonnance de statines. En 1996, année de l’arrivée de l’atorvastatine sur le marché, les parts de marché de la simvastatine et de la pravastatine étaient de 38,3 % et de 37,1 % respectivement. En 2003, la part de marché de l’atorvastatine était de 44 % par rapport à 29,9 % et à 24,1 % pour la simvastatine et la pravastatine respectivement. Par contre, la documentation publiée sur les ECH jusqu’à la fin de 2002 comptait 133 341 et 140 565 années-patient cumulées de suivi pour la simvastatine et la pravastatine respectivement contre seulement 1459 années-patient pour l’atorvastatine.
CONCLUSION: Les habitudes de prescription des médecins quant au choix des statines ne semblent pas uniquement déterminées par la bonne qualité des ECH. Aussi faudrait-il pousser plus loin la recherche sur les autres déterminants possibles des habitudes de prescription des médecins.
Figures

Comment in
-
Statin wars following coronary revascularization - evidence-based clinical practice?Can J Cardiol. 2007 Mar 15;23(4):319; author reply 319. doi: 10.1016/s0828-282x(07)70765-1. Can J Cardiol. 2007. PMID: 17407855 Free PMC article. No abstract available.
Similar articles
-
Low-density lipoprotein cholesterol (LDL-C) levels and LDL-C goal attainment among elderly patients treated with rosuvastatin compared with other statins in routine clinical practice.Am J Geriatr Pharmacother. 2007 Sep;5(3):185-94. doi: 10.1016/j.amjopharm.2007.10.002. Am J Geriatr Pharmacother. 2007. PMID: 17996658
-
Effectiveness of statins for secondary prevention in elderly patients after acute myocardial infarction: an evaluation of class effect.CMAJ. 2005 Apr 26;172(9):1187-94. doi: 10.1503/cmaj.1041403. CMAJ. 2005. PMID: 15851712 Free PMC article.
-
Class effects of statins in elderly patients with congestive heart failure: a population-based analysis.Am Heart J. 2008 Feb;155(2):316-23. doi: 10.1016/j.ahj.2007.09.004. Epub 2007 Oct 25. Am Heart J. 2008. PMID: 18215603
-
Are statins created equal? Evidence from randomized trials of pravastatin, simvastatin, and atorvastatin for cardiovascular disease prevention.Am Heart J. 2006 Feb;151(2):273-81. doi: 10.1016/j.ahj.2005.04.003. Am Heart J. 2006. PMID: 16442888 Review.
-
If LDL-C is the answer...what was the question? What do the data really show?Heart Dis. 2001 Jan-Feb;3(1):2-13. doi: 10.1097/00132580-200101000-00002. Heart Dis. 2001. PMID: 11975764 Review.
Cited by
-
Statin wars following coronary revascularization - evidence-based clinical practice?Can J Cardiol. 2007 Mar 15;23(4):319; author reply 319. doi: 10.1016/s0828-282x(07)70765-1. Can J Cardiol. 2007. PMID: 17407855 Free PMC article. No abstract available.
-
Curious Cases of the Enzymes.J Med Biochem. 2015 Jul;34(3):271-281. doi: 10.2478/jomb-2014-0045. Epub 2015 Jul 14. J Med Biochem. 2015. PMID: 28356837 Free PMC article. Review.
-
Adherence to statin treatment following a myocardial infarction: an Italian population-based survey.Clinicoecon Outcomes Res. 2015 Jun 1;7:273-80. doi: 10.2147/CEOR.S70936. eCollection 2015. Clinicoecon Outcomes Res. 2015. PMID: 26064062 Free PMC article.
References
-
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III) JAMA. 2001;285:2486–97. - PubMed
-
- The statin wars: Why AstraZeneca must retreat. Lancet. 2003;362:1341. - PubMed
-
- International Classification of Diseases. Ninth Revision. Geneva: World Health Organization; 1979.
-
- Conseil d’évaluation des technologies de la santé. Revascularization techniques used in the treatment of stable angina pectoris. Montréal: Conseil d’évaluation des technologies de la santé; 1996.
-
- Levy AR, Tamblyn RM, Fitchett D, McLeod PJ, Hanley JA. Coding accuracy of hospital discharge data for elderly survivors of myocardial infarction. Can J Cardiol. 1999;15:1277–82. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

