Loss of P4 ATPases Drs2p and Dnf3p disrupts aminophospholipid transport and asymmetry in yeast post-Golgi secretory vesicles
- PMID: 16452632
- PMCID: PMC1415292
- DOI: 10.1091/mbc.e05-10-0912
Loss of P4 ATPases Drs2p and Dnf3p disrupts aminophospholipid transport and asymmetry in yeast post-Golgi secretory vesicles
Abstract
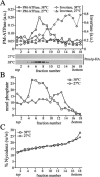
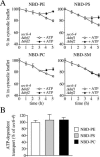
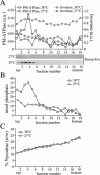
Eukaryotic plasma membranes generally display asymmetric lipid distributions with the aminophospholipids concentrated in the cytosolic leaflet. This arrangement is maintained by aminophospholipid translocases (APLTs) that use ATP hydrolysis to flip phosphatidylserine (PS) and phosphatidylethanolamine (PE) from the external to the cytosolic leaflet. The identity of APLTs has not been established, but prime candidates are members of the P4 subfamily of P-type ATPases. Removal of P4 ATPases Dnf1p and Dnf2p from budding yeast abolishes inward translocation of 6-[(7-nitrobenz-2-oxa-1,3-diazol-4-yl)aminocaproyl] (NBD)-labeled PS, PE, and phosphatidylcholine (PC) across the plasma membrane and causes cell surface exposure of endogenous PE. Here, we show that yeast post-Golgi secretory vesicles (SVs) contain a translocase activity that flips NBD-PS, NBD-PE, and NBD-PC to the cytosolic leaflet. This activity is independent of Dnf1p and Dnf2p but requires two other P4 ATPases, Drs2p and Dnf3p, that reside primarily in the trans-Golgi network. Moreover, SVs have an asymmetric PE arrangement that is lost upon removal of Drs2p and Dnf3p. Our results indicate that aminophospholipid asymmetry is created when membrane flows through the Golgi and that P4-ATPases are essential for this process.
Figures


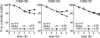


References
-
- Bell, R. M., Ballas, L. M., and Coleman, R. A. (1981). Lipid topogenesis. J. Lipid Res. 22, 391–403. - PubMed
-
- Birchmeier, W., Lanz, J. H., Winterhalter, K. H., and Conrad, M. J. (1979). ATP-induced endocytosis in human erythrocyte ghosts. Characterization of the process and isolation of the endocytosed vesicles. J. Biol. Chem. 254, 9298–9304. - PubMed
-
- Bishop, W. R., and Bell, R. M. (1985). Assembly of the endoplasmic reticulum phospholipid bilayer: the phosphatidylcholine transporter. Cell 42, 51–60. - PubMed
-
- Cribier, S., Morrot, G., and Zachowski, A. (1993). Dynamics of the membrane lipid phase. Prostaglandins Leukot. Essent. Fatty Acids 48, 27–32. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

