Use of electrochemical DNA biosensors for rapid molecular identification of uropathogens in clinical urine specimens
- PMID: 16455913
- PMCID: PMC1392664
- DOI: 10.1128/JCM.44.2.561-570.2006
Use of electrochemical DNA biosensors for rapid molecular identification of uropathogens in clinical urine specimens
Abstract
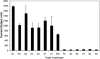
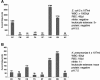
We describe the first species-specific detection of bacterial pathogens in human clinical fluid samples using a microfabricated electrochemical sensor array. Each of the 16 sensors in the array consisted of three single-layer gold electrodes-working, reference, and auxiliary. Each of the working electrodes contained one representative from a library of capture probes, each specific for a clinically relevant bacterial urinary pathogen. The library included probes for Escherichia coli, Proteus mirabilis, Pseudomonas aeruginosa, Enterocococcus spp., and the Klebsiella-Enterobacter group. A bacterial 16S rRNA target derived from single-step bacterial lysis was hybridized both to the biotin-modified capture probe on the sensor surface and to a second, fluorescein-modified detector probe. Detection of the target-probe hybrids was achieved through binding of a horseradish peroxidase (HRP)-conjugated anti-fluorescein antibody to the detector probe. Amperometric measurement of the catalyzed HRP reaction was obtained at a fixed potential of -200 mV between the working and reference electrodes. Species-specific detection of as few as 2,600 uropathogenic bacteria in culture, inoculated urine, and clinical urine samples was achieved within 45 min from the beginning of sample processing. In a feasibility study of this amperometric detection system using blinded clinical urine specimens, the sensor array had 100% sensitivity for direct detection of gram-negative bacteria without nucleic acid purification or amplification. Identification was demonstrated for 98% of gram-negative bacteria for which species-specific probes were available. When combined with a microfluidics-based sample preparation module, the integrated system could serve as a point-of-care device for rapid diagnosis of urinary tract infections.
Figures


References
-
- Abdel-Hamid, I., D. Ivnitski, P. Atanasov, and E. Wilkins. 1999. Flow-through immunofiltration assay system for rapid detection of E. coli O157:H7. Biosens. Bioelectron. 14:309-316. - PubMed
-
- Albers, J., T. Grunwald, E. Nebling, G. Piechotta, and R. Hintsche. 2003. Electrical biochip technology—a tool for microarrays and continuous monitoring. Anal. Bioanal. Chem. 377:521-527. - PubMed
-
- Bard, A. J., and L. R. Faulkner. 2001. Electrochemical methods fundamentals and applications, 2nd ed. John Wiley & Sons, Inc., Hoboken, N.J.
-
- Basu, M., S. Seggerson, J. Henshaw, J. Jiang, R. del A Cordona, C. Lefave, P. J. Boyle, A. Miller, M. Pugia, and S. Basu. 2004. Nano-biosensor development for bacterial detection during human kidney infection: use of glycoconjugate-specific antibody-bound gold NanoWire arrays (GNWA). Glycoconj. J. 21:487-496. - PubMed
-
- Campbell, C. N., D. Gal, N. Cristler, C. Banditrat, and A. Heller. 2002. Enzyme-amplified amperometric sandwich test for RNA and DNA. Anal. Chem. 74:158-162. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

