Results of the first World Health Organization international collaborative study of detection of human papillomavirus DNA
- PMID: 16455914
- PMCID: PMC1392673
- DOI: 10.1128/JCM.44.2.571-579.2006
Results of the first World Health Organization international collaborative study of detection of human papillomavirus DNA
Abstract
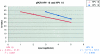
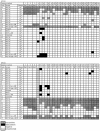
Twenty-nine laboratories in 12 countries participated in a study to assess the performance of various human papillomavirus (HPV) detection assays through the use of a recombinant HPV DNA standard reagent panel. The panel was designed by a group of HPV experts, and samples were prepared and distributed by the World Health Organization International Laboratory for Standards and Biologicals in The Netherlands. Each panel consisted of 24 coded samples including a dilution series for HPV types 16 and 18, alone or in combination with five other high-risk (HR) HPV types including HPV types 31, 33, 35, 45, and 52, the low-risk HPV type 6, and a negative control. Qualitative assays were generally consistent across laboratories, and most invalid results reflected a lack of HPV test sensitivity. The combined data sets had a proficiency for HPV 16 of 62.5% (15/24) and for HPV 18 of 73.9% (17/23). HPV 31 was the least accurately detected by participating laboratories. Approximately half of participating laboratories failed to detect high concentrations of HPV 31 and, to a lesser extent, to detect HPV types 35, 52, and 6. The panel sample materials offer a source of renewable and reproducible material that could be used in the future development of international standard reagents for calibration of HPV DNA assays and kits.
Figures
References
-
- Brown, D. R., J. T. Bryan, J. M. Schroeder, T. S. Robinson, K. H. Fife, C. M. Wheeler, E. Barr, P. R. Smith, L. Chiacchierini, A. DiCello, and K. U. Jansen. 2001. Neutralization of human papillomavirus type 11 (HPV-11) by serum from women vaccinated with yeast-derived HPV-11 L1 virus-like particles: correlation with competitive radioimmunoassay titer. J. Infect. Dis. 184:1183-1186. - PubMed
-
- de Villiers, E.-M., C. Fauquet, T. R. Broker, H.-U. Bernard, and H. zur Hausen. 2004. Classification of papillomaviruses. Virology 324:17-27. - PubMed
-
- Franco, E., L. L. Villa, T. Rohan, A. Ferenczy, M. Baggio, L. Galan, J. Robitaille, J. Sobrinho, A. Ruiz, J. Prado, M. Ribeiro, S. Ferreira, E. Duarte-Franco, J. Candeias, O. Caballero, P. Rahal, L. Sichero, M. Santos, M. Peltzl-Erler, and G. Matlashewski. 1999. Design and methods of the Ludwig-McGill longitudinal study of the natural history of human papillomavirus infection and cervical neoplasia in Brazil. Rev. Panam. Salud Publica 6:223-232. - PubMed
-
- Galloway, D. A. 2003. Papillomavirus vaccines in clinical trials. Lancet Infect. Dis. 3:469-475. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

