The progestational and androgenic properties of medroxyprogesterone acetate: gene regulatory overlap with dihydrotestosterone in breast cancer cells
- PMID: 16457685
- PMCID: PMC1410743
- DOI: 10.1186/bcr1340
The progestational and androgenic properties of medroxyprogesterone acetate: gene regulatory overlap with dihydrotestosterone in breast cancer cells
Abstract
Introduction: Medroxyprogesterone acetate (MPA), the major progestin used for oral contraception and hormone replacement therapy, has been implicated in increased breast cancer risk. Is this risk due to its progestational or androgenic properties? To address this, we assessed the transcriptional effects of MPA as compared with those of progesterone and dihydrotestosterone (DHT) in human breast cancer cells.
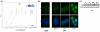
Method: A new progesterone receptor-negative, androgen receptor-positive human breast cancer cell line, designated Y-AR, was engineered and characterized. Transcription assays using a synthetic promoter/reporter construct, as well as endogenous gene expression profiling comparing progesterone, MPA and DHT, were performed in cells either lacking or containing progesterone receptor and/or androgen receptor.
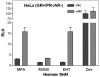
Results: In progesterone receptor-positive cells, MPA was found to be an effective progestin through both progesterone receptor isoforms in transient transcription assays. Interestingly, DHT signaled through progesterone receptor type B. Expression profiling of endogenous progesterone receptor-regulated genes comparing progesterone and MPA suggested that although MPA may be a somewhat more potent progestin than progesterone, it is qualitatively similar to progesterone. To address effects of MPA through androgen receptor, expression profiling was performed comparing progesterone, MPA and DHT using Y-AR cells. These studies showed extensive gene regulatory overlap between DHT and MPA through androgen receptor and none with progesterone. Interestingly, there was no difference between pharmacological MPA and physiological MPA, suggesting that high-dose therapeutic MPA may be superfluous.
Conclusion: Our comparison of the gene regulatory profiles of MPA and progesterone suggests that, for physiologic hormone replacement therapy, the actions of MPA do not mimic those of endogenous progesterone alone. Clinically, the complex pharmacology of MPA not only influences its side-effect profile; but it is also possible that the increased breast cancer risk and/or the therapeutic efficacy of MPA in cancer treatment is in part mediated by androgen receptor.
Figures
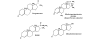

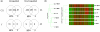

Similar articles
-
Anti-proliferative transcriptional effects of medroxyprogesterone acetate in estrogen receptor positive breast cancer cells are predominantly mediated by the progesterone receptor.J Steroid Biochem Mol Biol. 2020 May;199:105548. doi: 10.1016/j.jsbmb.2019.105548. Epub 2019 Dec 2. J Steroid Biochem Mol Biol. 2020. PMID: 31805393
-
A comparative study of the androgenic properties of progesterone and the progestins, medroxyprogesterone acetate (MPA) and norethisterone acetate (NET-A).J Steroid Biochem Mol Biol. 2014 Sep;143:404-15. doi: 10.1016/j.jsbmb.2014.05.007. Epub 2014 May 23. J Steroid Biochem Mol Biol. 2014. PMID: 24861265
-
Antiandrogenic actions of medroxyprogesterone acetate on epithelial cells within normal human breast tissues cultured ex vivo.Menopause. 2014 Jan;21(1):79-88. doi: 10.1097/GME.0b013e3182936ef4. Menopause. 2014. PMID: 23715406
-
Androgen receptor mediated growth control of breast cancer and endometrial cancer modulated by antiandrogen- and androgen-like steroids.J Steroid Biochem Mol Biol. 1996 Jan;56(1-6 Spec No):113-7. doi: 10.1016/0960-0760(95)00228-6. J Steroid Biochem Mol Biol. 1996. PMID: 8603031 Review.
-
Role of the androgen receptor in human breast cancer.J Mammary Gland Biol Neoplasia. 1998 Jan;3(1):95-103. doi: 10.1023/a:1018730519839. J Mammary Gland Biol Neoplasia. 1998. PMID: 10819508 Review.
Cited by
-
ALU repeats in promoters are position-dependent co-response elements (coRE) that enhance or repress transcription by dimeric and monomeric progesterone receptors.Mol Endocrinol. 2009 Jul;23(7):989-1000. doi: 10.1210/me.2009-0048. Epub 2009 Apr 16. Mol Endocrinol. 2009. PMID: 19372234 Free PMC article.
-
Progestin modulates the lipid profile and sensitivity of breast cancer cells to docetaxel.Mol Cell Endocrinol. 2012 Nov 5;363(1-2):111-21. doi: 10.1016/j.mce.2012.08.005. Epub 2012 Aug 16. Mol Cell Endocrinol. 2012. PMID: 22922095 Free PMC article.
-
Steroid hormones, steroid receptors, and breast cancer stem cells.J Mammary Gland Biol Neoplasia. 2015 Jun;20(1-2):39-50. doi: 10.1007/s10911-015-9340-5. Epub 2015 Aug 12. J Mammary Gland Biol Neoplasia. 2015. PMID: 26265122 Free PMC article. Review.
-
Sex and age influence gonadal steroid hormone receptor distributions relative to estrogen receptor β-containing neurons in the mouse hypothalamic paraventricular nucleus.J Comp Neurol. 2021 Jun;529(9):2283-2310. doi: 10.1002/cne.25093. Epub 2021 Jan 14. J Comp Neurol. 2021. PMID: 33341960 Free PMC article.
-
Unliganded progesterone receptors attenuate taxane-induced breast cancer cell death by modulating the spindle assembly checkpoint.Breast Cancer Res Treat. 2012 Jan;131(1):75-87. doi: 10.1007/s10549-011-1399-0. Epub 2011 Feb 22. Breast Cancer Res Treat. 2012. PMID: 21340479 Free PMC article.
References
-
- Piccinino LJ, Mosher WD. Trends in contraceptive use in the United States: 1982–1995. Fam Plann Perspect. 1998;30:4–10. 46. - PubMed
-
- Shumaker SA, Legault C, Rapp SR, Thal L, Wallace RB, Ockene JK, Hendrix SL, Jones BN, 3rd, Assaf AR, Jackson RD, et al. Estrogen plus progestin and the incidence of dementia and mild cognitive impairment in postmenopausal women: the Women's Health Initiative Memory Study: a randomized controlled trial. JAMA. 2003;289:2651–2662. doi: 10.1001/jama.289.20.2651. - DOI - PubMed
-
- Rossouw JE, Anderson GL, Prentice RL, LaCroix AZ, Kooperberg C, Stefanick ML, Jackson RD, Beresford SA, Howard BV, Johnson KC, Writing Group for the Women's Health Initiative Investigators et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women's Health Initiative randomized controlled trial. JAMA. 2002;288:321–333. doi: 10.1001/jama.288.3.321. - DOI - PubMed
-
- Anderson GL, Limacher M, Assaf AR, Bassford T, Beresford SA, Black H, Bonds D, Brunner R, Brzyski R, Caan B, et al. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy: the Women's Health Initiative randomized controlled trial. JAMA. 2004;291:1701–1712. doi: 10.1001/jama.291.14.1701. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

