Prognostic significance of pathological response after neoadjuvant chemotherapy or chemoradiation for locally advanced cervical carcinoma
- PMID: 16457727
- PMCID: PMC1386679
- DOI: 10.1186/1477-7800-3-3
Prognostic significance of pathological response after neoadjuvant chemotherapy or chemoradiation for locally advanced cervical carcinoma
Abstract
Background: Cisplatin-based chemoradiation is the standard of care for locally advanced cervical cancer patients; however, neoadjuvant modalities are currently being tested. Neoadjuvant studies in several tumor types have underscored the prognostic significance of pathological response for survival; however there is a paucity of studies in cervical cancer investigating this issue.
Methods: Four cohorts of patients with locally advanced cervical carcinoma (stages IB2-IIIB); included prospectively in phase II protocols of either neoadjuvant chemotherapy with 1) cisplatin-gemcitabine, 2) oxaliplatin-gemcitabine, 3) carboplatin-paclitaxel or 4) chemoradiation with cisplatin or cisplatin-gemcitabine followed by radical hysterectomy were analyzed for pathological response and survival.
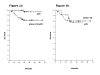
Results: One-hundred and fifty three (86%) of the 178 patients treated within these trials, underwent radical hysterectomy and were analyzed. Overall, the mean age was 44.7 and almost two-thirds were FIGO stage IIB. Pathological response rates were as follows: Complete (pCR) in 60 cases (39.2%), Near-complete (p-Near-CR) in 24 (15.6 %) and partial (pPR) in 69 cases (45.1%). A higher proportion rate of pCR was observed in patients treated with chemoradiotherapy (with cisplatin [19/40, 47.5%]; or with cisplatin-gemcitabine [24/41, 58.5%] compared with patients receiving only chemotherapy, 6/23 (26%), 3/8 (37.5%) and 8/41 (19.5%) for cisplatin-gemcitabine, oxaliplatin-gemcitabine and carboplatin-paclitaxel respectively [p = 0.0001]). A total of 29 relapses (18.9%) were documented. The pathological response was the only factor influencing on relapse, since only 4/60 (6.6%) patients with pCR relapsed, compared with 25/93 (26.8%) patients with viable tumor, either pNear-CR or pPR (p = 0.001). Overall survival was 98.3% in patients with pCR versus 83% for patients with either pNear-CR or pPR (p = 0.009).
Conclusion: Complete pathological response but no Near-complete and partial responses is associated with longer survival in cervical cancer patients treated with neoadjuvant chemotherapy or chemoradiotherapy.
Figures
References
-
- Mohar A, Frias-Mendivil M. Epidemiology of cervical cancer. Cancer Invest. 2000;18:584–590. - PubMed
-
- Peters WA, 3rd, Liu PY, Barrett R, Stock RJ, Monk BJ, Berek JS, Souhami L, Grigsby P, Gordon W, Jr, Alberts DS. Concurrent chemotherapy and pelvic radiation therapy compared with pelvic radiation therapy alone as adjuvant therapy after radical surgery in high-risk early-stage cancer of the cervix. J Clin Oncol. 2000;18:1606–1613. - PubMed
-
- Morris M, Eifel PJ, Lu J, Grigsby PW, Levenback C, Stevens RE, Rotman M, Gerhenson DM, Mutch DG. Pelvic radiation with concurrent chemotherapy compared with pelvic and para-aortic radiation for high-risk cervical cancer. New Engl J Med. 1999;340:1137–1143. doi: 10.1056/NEJM199904153401501. - DOI - PubMed
-
- Keys HM, Bundy BN, Stehman FB, Muderspach LI, Chafe WE, Suggs CL, Walker JL, Gersell D. A comparison of weekly cisplatin during radiation therapy versus irradiation alone each followed by adjuvant hysterectomy in bulky stage IB cervical carcinoma: a randomized trial of the Gynecology Oncology Group. New Engl J Med. 1999;340:1154–1161. doi: 10.1056/NEJM199904153401503. - DOI - PubMed
-
- Rose PG, Bundy BN, Watkins EB, Thigpen JT, Deppe G, Maiman MA, Clarke-Pearson DL, Insalacos S. Concurrent cisplatin-based chemoradiation improves progression-free survival in advanced cervical cancer: results of a randomized Gynecologic Oncology Group study. New Engl J Med. 1999;340:1144–1153. doi: 10.1056/NEJM199904153401502. - DOI - PubMed
LinkOut - more resources
Full Text Sources

