Calpain is required for normal osteoclast function and is down-regulated by calcitonin
- PMID: 16461769
- PMCID: PMC1570620
- DOI: 10.1074/jbc.M513516200
Calpain is required for normal osteoclast function and is down-regulated by calcitonin
Abstract
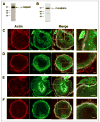
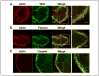
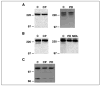
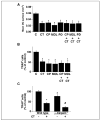
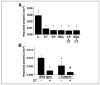
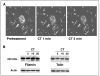
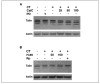
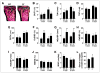
Osteoclast motility is thought to depend on rapid podosome assembly and disassembly. Both mu-calpain and m-calpain, which promote the formation and disassembly of focal adhesions, were observed in the podosome belt of osteoclasts. Calpain inhibitors disrupted the podosome belt, blocked the constitutive cleavage of the calpain substrates filamin A, talin, and Pyk2, which are enriched in the podosome belt, induced osteoclast retraction, and reduced osteoclast motility and bone resorption. The motility and resorbing activity of mu-calpain(-/-) osteoclast-like cells were also reduced, indicating that mu-calpain is required for normal osteoclast activity. Histomorphometric analysis of tibias from mu-calpain(-/-) mice revealed increased osteoclast numbers and decreased trabecular bone volume that was apparent at 10 weeks but not at 5 weeks of age. In vitro studies suggested that the increased osteoclast number in the mu-calpain(-/-) bones resulted from increased osteoclast survival, not increased osteoclast formation. Calcitonin disrupted the podosome ring, induced osteoclast retraction, and reduced osteoclast motility and bone resorption in a manner similar to the effects of calpain inhibitors and had no further effect on these parameters when added to osteoclasts pretreated with calpain inhibitors. Calcitonin inhibited the constitutive cleavage of a fluorogenic calpain substrate and transiently blocked the constitutive cleavage of filamin A, talin, and Pyk2 by a protein kinase C-dependent mechanism, demonstrating that calcitonin induces the inhibition of calpain in osteoclasts. These results indicate that calpain activity is required for normal osteoclast activity and suggest that calcitonin inhibits osteoclast bone resorbing activity in part by down-regulating calpain activity.
Figures
Similar articles
-
Calcitonin inhibits SDCP-induced osteoclast apoptosis and increases its efficacy in a rat model of osteoporosis.PLoS One. 2012;7(7):e40272. doi: 10.1371/journal.pone.0040272. Epub 2012 Jul 6. PLoS One. 2012. PMID: 22792258 Free PMC article.
-
Calcitonin induces podosome disassembly and detachment of osteoclasts by modulating Pyk2 and Src activities.Bone. 2007 May;40(5):1329-42. doi: 10.1016/j.bone.2007.01.014. Epub 2007 Jan 26. Bone. 2007. PMID: 17321230
-
Functional osteoclast attachment requires inositol-1,4,5-trisphosphate receptor-associated cGMP-dependent kinase substrate.Lab Invest. 2010 Oct;90(10):1533-42. doi: 10.1038/labinvest.2010.120. Epub 2010 Jun 21. Lab Invest. 2010. PMID: 20567233 Free PMC article.
-
[Effects of calcitonin on osteoclast].Clin Calcium. 2005 Mar;15(3):147-51. Clin Calcium. 2005. PMID: 15741694 Review. Japanese.
-
Osteoclast function and its control.Exp Physiol. 1993 Nov;78(6):721-39. doi: 10.1113/expphysiol.1993.sp003721. Exp Physiol. 1993. PMID: 8311941 Review.
Cited by
-
Macrophage mesenchymal migration requires podosome stabilization by filamin A.J Biol Chem. 2012 Apr 13;287(16):13051-62. doi: 10.1074/jbc.M111.307124. Epub 2012 Feb 9. J Biol Chem. 2012. PMID: 22334688 Free PMC article.
-
Calcitonin inhibits SDCP-induced osteoclast apoptosis and increases its efficacy in a rat model of osteoporosis.PLoS One. 2012;7(7):e40272. doi: 10.1371/journal.pone.0040272. Epub 2012 Jul 6. PLoS One. 2012. PMID: 22792258 Free PMC article.
-
Cortactin has an essential and specific role in osteoclast actin assembly.Mol Biol Cell. 2006 Jul;17(7):2882-95. doi: 10.1091/mbc.e06-03-0187. Epub 2006 Apr 12. Mol Biol Cell. 2006. PMID: 16611741 Free PMC article.
-
Fluid shear stress and sphingosine 1-phosphate activate calpain to promote membrane type 1 matrix metalloproteinase (MT1-MMP) membrane translocation and endothelial invasion into three-dimensional collagen matrices.J Biol Chem. 2011 Dec 9;286(49):42017-42026. doi: 10.1074/jbc.M111.290841. Epub 2011 Oct 14. J Biol Chem. 2011. PMID: 22002053 Free PMC article.
-
Augmenting MNK1/2 activation by c-FMS proteolysis promotes osteoclastogenesis and arthritic bone erosion.Bone Res. 2021 Oct 20;9(1):45. doi: 10.1038/s41413-021-00162-0. Bone Res. 2021. PMID: 34671034 Free PMC article.
References
-
- Tarone G, Cirillo D, Giancotti FG, Comoglio PM, Marchisio PC. Exp Cell Res. 1985;159:141–157. - PubMed
-
- Marchisio PC, Cirillo D, Teti A, Zambonin-Zallone A, Tarone G. Exp Cell Res. 1987;169:202–214. - PubMed
-
- Nermut MV, Eason P, Hirst EMA, Kellie S. Exp Cell Res. 1991;193:382–397. - PubMed
-
- Pfaff M, Jurdic P. J Cell Sci. 2001;114:2775–2786. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 HL051445/HL/NHLBI NIH HHS/United States
- R01 DE004724/DE/NIDCR NIH HHS/United States
- AR-49879/AR/NIAMS NIH HHS/United States
- R56 AR049879/AR/NIAMS NIH HHS/United States
- R01 AR057769/AR/NIAMS NIH HHS/United States
- P30 AR046032/AR/NIAMS NIH HHS/United States
- R37 DE004724/DE/NIDCR NIH HHS/United States
- DE-04724/DE/NIDCR NIH HHS/United States
- R01 HL089517/HL/NHLBI NIH HHS/United States
- HL-51445/HL/NHLBI NIH HHS/United States
- R56 HL051445/HL/NHLBI NIH HHS/United States
- AR-46032/AR/NIAMS NIH HHS/United States
- R01 AR049879/AR/NIAMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

