Block of inferior olive gap junctional coupling decreases Purkinje cell complex spike synchrony and rhythmicity
- PMID: 16467522
- PMCID: PMC6793617
- DOI: 10.1523/JNEUROSCI.3677-05.2006
Block of inferior olive gap junctional coupling decreases Purkinje cell complex spike synchrony and rhythmicity
Abstract
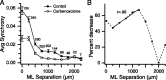
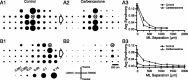
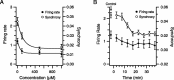
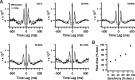
Inferior olivary (IO) neurons are electrotonically coupled by gap junctions. This coupling is thought to underlie synchronous complex spike (CS) activity generated by the olivocerebellar system in Purkinje cells, and also has been hypothesized to be necessary for IO neurons to generate spontaneous oscillatory activity. These characteristics of olivocerebellar activity have been proposed to be central to the role of this system in motor coordination. However, the relationship of gap junction coupling between IO neurons to synchronous and rhythmic CS activity has never been directly tested. Thus, to address this issue, multiple electrode recordings were obtained from crus 2a Purkinje cells, and carbenoxolone, a gap junction blocker, was injected into the IO. Carbenoxolone reduced CS synchrony by 50% overall, but in some experiments, >80% reductions were achieved. Carbenoxolone also reduced the average firing rate by 50%, suggesting that electrical coupling is a significant source of excitation for IO neurons. Moreover, carbenoxolone caused a reduction in the approximately 10 Hz rhythmicity of CS activity, and this reduction was correlated with the extent to which the injection reduced CS synchrony. Lastly, carbenoxolone was found to reverse or prevent changes in synchrony that are normally induced by injection of GABAA and glutamate receptor antagonists into the IO, suggesting that the effects of these drugs on CS synchrony patterns require electrical coupling of IO neurons. In sum, our results provide direct evidence that electrical coupling of IO neurons underlies synchronous CS activity, and suggest important roles for this coupling in shaping other aspects of IO spiking patterns.
Figures





References
-
- Bal T, McCormick DA (1997). Synchronized oscillations in the inferior olive are controlled by the hyperpolarization-activated cation current Ih J Neurophysiol 77:3145–3156. - PubMed
-
- Bell CC, Kawasaki T (1972). Relations among climbing fiber responses of nearby Purkinje cells. J Neurophysiol 35:155–169. - PubMed
-
- Belluardo N, Mudò G, Travato-Salinaro A, Le Gurun S, Charollais A, Serre-Beinier V, Amato G, Haefliger J-A, Meda P, Condorelli DF (2000). Expression of connexin36 in the adult and developing rat brain. Brain Res 865:121–138. - PubMed
-
- Benardo LS, Foster RE (1986). Oscillatory behavior in inferior olive neurons: mechanisms, modulation, cell aggregates. Brain Res Bull 17:773–784. - PubMed
-
- Bleasel AF, Pettigrew AG (1992). Development and properties of spontaneous oscillations of the membrane potential in inferior olivary neurons in the rat. Brain Res Dev Brain Res 65:43–50. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
