Association between hepatitis C virus and very-low-density lipoprotein (VLDL)/LDL analyzed in iodixanol density gradients
- PMID: 16474148
- PMCID: PMC1395398
- DOI: 10.1128/JVI.80.5.2418-2428.2006
Association between hepatitis C virus and very-low-density lipoprotein (VLDL)/LDL analyzed in iodixanol density gradients
Abstract
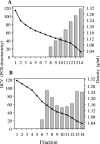
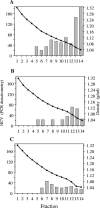
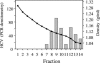
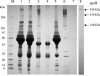
Hepatitis C virus (HCV) RNA circulates in the blood of persistently infected patients in lipoviroparticles (LVPs), which are heterogeneous in density and associated with host lipoproteins and antibodies. The variability and lability of these virus-host complexes on fractionation has hindered our understanding of the structure of LVP and determination of the physicochemical properties of the HCV virion. In this study, HCV from an antibody-negative immunodeficient patient was analyzed using three fractionation techniques, NaBr gradients, isotonic iodixanol, and sucrose gradient centrifugation. Iodixanol gradients were shown to best preserve host lipoprotein-virus complexes, and all HCV RNA was found at densities below 1.13 g/ml, with the majority at low density, < or =1.08 g/ml. Immunoprecipitation with polyclonal antibodies against human ApoB and ApoE precipitated 91.8% and 95.0% of HCV with low density, respectively, suggesting that host lipoprotein is closely associated with HCV in a particle resembling VLDL. Immunoprecipitation with antibodies against glycoprotein E2 precipitated 25% of HCV with low density, providing evidence for the presence of E2 in LVPs. Treatment of serum with 0.5% deoxycholic acid in the absence of salt produced HCV with a density of 1.12 g/ml and a sedimentation coefficient of 215S. The diameters of these particles were calculated as 54 nm. Treatment of serum with 0.18% NP-40 produced HCV with a density of 1.18 g/ml, a sedimentation coefficient of 180S, and a diameter of 42 nm. Immunoprecipitation analysis showed that ApoB remained associated with HCV after treatment of serum with deoxycholic acid or NP-40, whereas ApoE was removed from HCV with these detergents.
Figures


References
-
- Agnello, V., R. T. Chung, and L. M. Kaplan. 1992. A role for hepatitis C virus infection in type II cryoglobulinemia. N. Engl. J. Med. 327:1490-1495. - PubMed
-
- André, P., G. Perlemuter, A. Budkowska, C. Bréchot, and V. Lotteau. 2005. Hepatitis C virus particles and lipoprotein metabolism. Semin. Liver Dis. 25:93-104. - PubMed
-
- Andrews, A. T. 1989. Electrophoresis. Theory, techniques, and biochemical and clinical applications, 2nd ed., p. 93-116. Oxford University Press, Oxford, England.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

