Kappa opioids selectively control dopaminergic neurons projecting to the prefrontal cortex
- PMID: 16477003
- PMCID: PMC1413839
- DOI: 10.1073/pnas.0511159103
Kappa opioids selectively control dopaminergic neurons projecting to the prefrontal cortex
Abstract
Dopaminergic afferents arising from the ventral tegmental area (VTA) are crucial elements in the neural circuits that mediate arousal, motivation, and reinforcement. Two major targets of these afferents are the medial prefrontal cortex (mPFC) and the nucleus accumbens (NAc). Whereas dopamine (DA) in the mPFC has been implicated in working memory and attentional processes, DA in the NAc is required for responding to reward predictive cues. These distinct functions suggest a role for independent firing patterns of dopaminergic neurons projecting to these brain regions. In fact, DA release in mPFC and NAc can be differentially modulated. However, to date, electrophysiological studies have largely overlooked heterogeneity among VTA neurons. Here, we provide direct evidence for differential neurotransmitter control of DA neural activity and corresponding DA release based on projection target. Kappa opioid receptor agonists inhibit VTA DA neurons that project to the mPFC but not those that project to the NAc. Moreover, DA levels in the mPFC, but not the NAc, are reduced after local infusion of kappa opioid receptor agonists into the VTA. These findings demonstrate that DA release in specific brain regions can be independently regulated by opioid targeting of a subpopulation of VTA DA neurons. Selective control of VTA DA neurons projecting to the mPFC has important implications for understanding addiction, attention disorders, and schizophrenia, all of which are associated with DA dysfunction in the mPFC.
Conflict of interest statement
Conflict of interest statement: No conflicts declared.
Figures

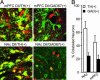
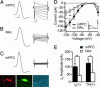
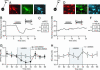
References
-
- McClure S. M., Daw N. D., Montague P. R. Trends Neurosci. 2003;26:423–428. - PubMed
-
- Schultz W. Neuron. 2002;36:241–263. - PubMed
-
- Wise R. A. Nat. Rev. Neurosci. 2004;5:483–494. - PubMed
-
- Williams G. V., Goldman-Rakic P. S. Nature. 1995;376:572–575. - PubMed
-
- Chudasama Y., Robbins T. W. Psychopharmacology. 2004;174:86–98. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

