Acceleration of age-related hearing loss by early noise exposure: evidence of a misspent youth
- PMID: 16481444
- PMCID: PMC1855187
- DOI: 10.1523/JNEUROSCI.4985-05.2006
Acceleration of age-related hearing loss by early noise exposure: evidence of a misspent youth
Abstract
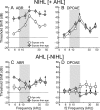
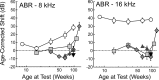
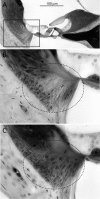
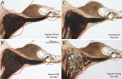
Age-related and noise-induced hearing losses in humans are multifactorial, with contributions from, and potential interactions among, numerous variables that can shape final outcome. A recent retrospective clinical study suggests an age-noise interaction that exacerbates age-related hearing loss in previously noise-damaged ears (Gates et al., 2000). Here, we address the issue in an animal model by comparing noise-induced and age-related hearing loss (NIHL; AHL) in groups of CBA/CaJ mice exposed identically (8-16 kHz noise band at 100 dB sound pressure level for 2 h) but at different ages (4-124 weeks) and held with unexposed cohorts for different postexposure times (2-96 weeks). When evaluated 2 weeks after exposure, maximum threshold shifts in young-exposed animals (4-8 weeks) were 40-50 dB; older-exposed animals (> or =16 weeks) showed essentially no shift at the same postexposure time. However, when held for long postexposure times, animals with previous exposure demonstrated AHL and histopathology fundamentally unlike unexposed, aging animals or old-exposed animals held for 2 weeks only. Specifically, they showed substantial, ongoing deterioration of cochlear neural responses, without additional change in preneural responses, and corresponding histologic evidence of primary neural degeneration throughout the cochlea. This was true particularly for young-exposed animals; however, delayed neuropathy was observed in all noise-exposed animals held 96 weeks after exposure, even those that showed no NIHL 2 weeks after exposure. Data suggest that pathologic but sublethal changes initiated by early noise exposure render the inner ears significantly more vulnerable to aging.
Figures


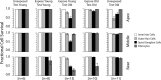
References
-
- Adams JC (2002). Clinical implications of inflammatory cytokines in the cochlea: a technical note. Otol Neurotol 23:316–322. - PubMed
-
- American College of Occupational Medicine. (1989). Occupational noise-induced hearing loss. ACOM Noise and Hearing Conservation Committee. J Occup Med 31:996–1001. - PubMed
-
- Bock GR, Saunders JC (1977). A critical period for acoustic trauma in the hamster and its relation to cochlear development. Science 197:396–398. - PubMed
-
- Bock GR, Seifter EJ (1978). Developmental changes of susceptibility to auditory fatigue in young hamsters. Audiology 17:193–203. - PubMed
-
- Candreia C, Martin GK, Stagner BB, Lonsbury-Martin BL (2004). Distortion product otoacoustic emissions show exceptional resistance to noise exposure in MOLF/Ei mice. Hear Res 194:109–117. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
