Dodecaprenyl phosphate-galacturonic acid as a donor substrate for lipopolysaccharide core glycosylation in Rhizobium leguminosarum
- PMID: 16497671
- PMCID: PMC2556281
- DOI: 10.1074/jbc.M513865200
Dodecaprenyl phosphate-galacturonic acid as a donor substrate for lipopolysaccharide core glycosylation in Rhizobium leguminosarum
Abstract
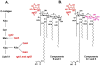
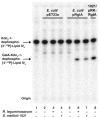
The lipid A and inner core regions of Rhizobium leguminosarum lipopolysaccharide contain four galacturonic acid (GalA) residues. Two are attached to the outer unit of the 3-deoxy-D-manno-octulosonic acid (Kdo) disaccharide, one to the mannose residue, and one to the 4'-position of lipid A. The enzymes RgtA and RgtB, described in the accompanying article, catalyze GalA transfer to the Kdo residue, whereas RgtC is responsible for modification of the core mannose unit. Heterologous expression of RgtA in Sinorhizhobium meliloti 1021, a strain that normally lacks GalA modifications on its Kdo disaccharide, resulted in detectable GalA transferase activity in isolated membrane preparations, suggesting that the appropriate GalA donor substrate is available in S. meliloti membranes. In contrast, heterologous expression of RgtA in Escherichia coli yielded inactive membranes. However, RgtA activity was detectable in the E. coli system when total lipids from R. leguminosarum 3841 or S. meliloti 1021 were added. We have now purified and characterized dodecaprenyl (C60) phosphate-GalA as a minor novel lipid of R. leguminosarum 3841 and S. meliloti. This substance is stable to mild base hydrolysis and was purified by DEAE-cellulose column chromatography. Its structure was established by a combination of electrospray ionization mass spectrometry and gas-liquid chromatography. Purified dodecaprenyl phosphate-GalA supports the efficient transfer of GalA to Kdo2-1-dephospho-lipid IV(A) by membranes of E. coli cells expressing RgtA, RgtB, and RgtC. The identification of a polyisoprene phosphate-GalA donor substrate suggests that the active site of RgtA faces the periplasmic side of the inner membrane. This work represents the first definitive characterization of a lipid-linked GalA derivative with the proposed structure dodecaprenyl phosphate-beta-D-GalA.
Figures







References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

