Constitutive NF-kappaB and NFAT activation leads to stimulation of the BLyS survival pathway in aggressive B-cell lymphomas
- PMID: 16497967
- PMCID: PMC1895801
- DOI: 10.1182/blood-2005-10-4042
Constitutive NF-kappaB and NFAT activation leads to stimulation of the BLyS survival pathway in aggressive B-cell lymphomas
Abstract
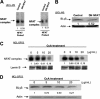
B-lymphocyte stimulator (BLyS), a relatively recently recognized member of the tumor necrosis factor ligand family (TNF), is a potent cell-survival factor expressed in many hematopoietic cells. BLyS binds to 3 TNF-R receptors, TACI, BCMA, BAFF-R, to regulate B-cell survival, differentiation, and proliferation. The mechanisms involved in BLYS gene expression and regulation are still incompletely understood. In this study, we examined BLYS gene expression, function, and regulation in B-cell non-Hodgkin lymphoma (NHL-B) cells. Our studies indicate that BLyS is constitutively expressed in aggressive NHL-B cells, including large B-cell lymphoma (LBCL) and mantle cell lymphoma (MCL), playing an important role in the survival and proliferation of malignant B cells. We found that 2 important transcription factors, NF-kappaB and NFAT, are involved in regulating BLyS expression through at least one NF-kappaB and 2 NFAT binding sites in the BLYS promoter. We also provide evidence suggesting that the constitutive activation of NF-kappaB and BLyS in NHL-B cells forms a positive feedback loop associated with lymphoma cell survival and proliferation. Our findings indicate that constitutive NF-kappaB and NFAT activations are crucial transcriptional regulators of the BLyS survival pathway in malignant B cells that could be therapeutic targets in aggressive NHL-B.
Figures




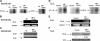

Similar articles
-
Lymphoma B cells evade apoptosis through the TNF family members BAFF/BLyS and APRIL.J Immunol. 2004 Mar 1;172(5):3268-79. doi: 10.4049/jimmunol.172.5.3268. J Immunol. 2004. PMID: 14978135
-
Expression of BLyS and its receptors in B-cell non-Hodgkin lymphoma: correlation with disease activity and patient outcome.Blood. 2004 Oct 15;104(8):2247-53. doi: 10.1182/blood-2004-02-0762. Epub 2004 Jul 13. Blood. 2004. PMID: 15251985
-
Constitutive NF-kappaB and NFAT activation in aggressive B-cell lymphomas synergistically activates the CD154 gene and maintains lymphoma cell survival.Blood. 2005 Dec 1;106(12):3940-7. doi: 10.1182/blood-2005-03-1167. Epub 2005 Aug 11. Blood. 2005. PMID: 16099873 Free PMC article.
-
Role of BAFF/BAFF-R axis in B-cell non-Hodgkin lymphoma.Crit Rev Oncol Hematol. 2014 Aug;91(2):113-22. doi: 10.1016/j.critrevonc.2014.02.004. Epub 2014 Feb 23. Crit Rev Oncol Hematol. 2014. PMID: 24629840 Review.
-
Mechanism of BLyS action in B cell immunity.Cytokine Growth Factor Rev. 2002 Feb;13(1):19-25. doi: 10.1016/s1359-6101(01)00025-9. Cytokine Growth Factor Rev. 2002. PMID: 11750877 Review.
Cited by
-
Systems biology of lupus: mapping the impact of genomic and environmental factors on gene expression signatures, cellular signaling, metabolic pathways, hormonal and cytokine imbalance, and selecting targets for treatment.Autoimmunity. 2010 Feb;43(1):32-47. doi: 10.3109/08916930903374774. Autoimmunity. 2010. PMID: 20001421 Free PMC article. Review.
-
Bacterial Lipoproteins Induce BAFF Production via TLR2/MyD88/JNK Signaling Pathways in Dendritic Cells.Front Immunol. 2020 Oct 2;11:564699. doi: 10.3389/fimmu.2020.564699. eCollection 2020. Front Immunol. 2020. PMID: 33123136 Free PMC article.
-
NFAT2 Isoforms Differentially Regulate Gene Expression, Cell Death, and Transformation through Alternative N-Terminal Domains.Mol Cell Biol. 2015 Oct 19;36(1):119-31. doi: 10.1128/MCB.00501-15. Print 2016 Jan 1. Mol Cell Biol. 2015. PMID: 26483414 Free PMC article.
-
Expression of BAFF and BAFF-R in follicular lymphoma: correlation with clinicopathologic characteristics and survival outcomes.PLoS One. 2012;7(12):e50936. doi: 10.1371/journal.pone.0050936. Epub 2012 Dec 13. PLoS One. 2012. PMID: 23272079 Free PMC article.
-
Differential effects on BAFF and APRIL levels in rituximab-treated patients with systemic lupus erythematosus and rheumatoid arthritis.Arthritis Res Ther. 2006;8(6):R167. doi: 10.1186/ar2076. Arthritis Res Ther. 2006. PMID: 17092341 Free PMC article. Clinical Trial.
References
-
- Moore PA, Belvedere O, Orr A, et al. BLyS: member of the tumor necrosis factor family and B lymphocyte stimulater. Science. 1999;285: 260-263. - PubMed
-
- Mukhopadhyay A, Ni J, Zhai Y, Yu GL, Aggarwal BB. Identification and characterization of a novel cytokine, THANK, a TNF homologue that activate apoptosis, nuclear factor-kappaB, and c-Jun NH2-terminal kinase. J Biol Chem. 1999;274: 15978-15981. - PubMed
-
- Shu HB, Hu WH, Johnson H. TALL-1 is a novel member of the TNF family that is down-regulated by mitogens. J Leukoc Biol. 1999;65: 680-683. - PubMed
-
- Batten M, Fletcher C, Ng LG, et al. TNF deficiency fails to protect BAFF transgenic mice against autoimmunity and reveals a prediction to B cell lymphoma. J Immunol. 2004;172: 812-822. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

