IgG-blocking antibodies inhibit IgE-mediated anaphylaxis in vivo through both antigen interception and Fc gamma RIIb cross-linking
- PMID: 16498503
- PMCID: PMC1378186
- DOI: 10.1172/JCI25575
IgG-blocking antibodies inhibit IgE-mediated anaphylaxis in vivo through both antigen interception and Fc gamma RIIb cross-linking
Abstract
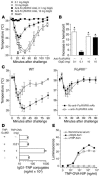
Although it has long been hypothesized that allergen immunotherapy inhibits allergy, in part, by inducing production of IgG Abs that intercept allergens before they can cross-link mast cell Fc epsilonRI-associated IgE, this blocking Ab hypothesis has never been tested in vivo. In addition, evidence that IgG-allergen interactions can induce anaphylaxis by activating macrophages through Fc gammaRIII suggested that IgG Ab might not be able to inhibit IgE-mediated anaphylaxis without inducing anaphylaxis through this alternative pathway. We have studied active and passive immunization models in mice to approach these issues and to determine whether any inhibition of anaphylaxis observed was a direct effect of allergen neutralization by IgG Ab or an indirect effect of cross-linking of Fc epsilonRI to the inhibitory IgG receptor Fc gammaRIIb. We demonstrate that IgG Ab produced during the course of an immune response or administered passively can completely suppress IgE-mediated anaphylaxis; that these IgG blocking Abs inhibit IgE-mediated anaphylaxis without inducing Fc gammaRIII-mediated anaphylaxis only when IgG Ab concentration is high and challenge allergen dose is low; that allergen epitope density correlates inversely with the allergen dose required to induce both IgE- and Fc gammaRIII-mediated anaphylaxis; and that both allergen interception and Fc gammaRIIb-dependent inhibition contribute to in vivo blocking Ab activity.
Figures


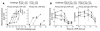

Similar articles
-
Absence of Fc epsilonRI alpha chain results in upregulation of Fc gammaRIII-dependent mast cell degranulation and anaphylaxis. Evidence of competition between Fc epsilonRI and Fc gammaRIII for limiting amounts of FcR beta and gamma chains.J Clin Invest. 1997 Mar 1;99(5):915-25. doi: 10.1172/JCI119256. J Clin Invest. 1997. PMID: 9062349 Free PMC article.
-
Systemic anaphylaxis in the mouse can be mediated largely through IgG1 and Fc gammaRIII. Assessment of the cardiopulmonary changes, mast cell degranulation, and death associated with active or IgE- or IgG1-dependent passive anaphylaxis.J Clin Invest. 1997 Mar 1;99(5):901-14. doi: 10.1172/JCI119255. J Clin Invest. 1997. PMID: 9062348 Free PMC article.
-
IgG-mediated anaphylaxis via Fc gamma receptor in CD40-deficient mice.Clin Exp Immunol. 1998 Nov;114(2):154-60. doi: 10.1046/j.1365-2249.1998.00717.x. Clin Exp Immunol. 1998. PMID: 9822270 Free PMC article.
-
Molecular mechanisms of anaphylaxis: lessons from studies with murine models.J Allergy Clin Immunol. 2005 Mar;115(3):449-57; quiz 458. doi: 10.1016/j.jaci.2004.12.1125. J Allergy Clin Immunol. 2005. PMID: 15753886 Review.
-
IgE and IgG Antibodies as Regulators of Mast Cell and Basophil Functions in Food Allergy.Front Immunol. 2020 Dec 11;11:603050. doi: 10.3389/fimmu.2020.603050. eCollection 2020. Front Immunol. 2020. PMID: 33362785 Free PMC article. Review.
Cited by
-
Differential roles for the IL-9/IL-9 receptor alpha-chain pathway in systemic and oral antigen-induced anaphylaxis.J Allergy Clin Immunol. 2010 Feb;125(2):469-476.e2. doi: 10.1016/j.jaci.2009.09.054. J Allergy Clin Immunol. 2010. PMID: 20159257 Free PMC article.
-
Interleukin-10-secreting regulatory T cells in allergy and asthma.Curr Allergy Asthma Rep. 2006 Sep;6(5):363-71. doi: 10.1007/s11882-996-0005-8. Curr Allergy Asthma Rep. 2006. PMID: 16899197 Review.
-
Research Advances in Mast Cell Biology and Their Translation Into Novel Therapies for Anaphylaxis.J Allergy Clin Immunol Pract. 2023 Jul;11(7):2032-2042. doi: 10.1016/j.jaip.2023.03.015. Epub 2023 Mar 21. J Allergy Clin Immunol Pract. 2023. PMID: 36958519 Free PMC article. Review.
-
Importance of mast cell histamine secretion in IgG-mediated systemic anaphylaxis.J Allergy Clin Immunol. 2025 Mar;155(3):956-973. doi: 10.1016/j.jaci.2024.11.009. Epub 2024 Nov 15. J Allergy Clin Immunol. 2025. PMID: 39550048
-
Pathogenic and Nonpathogenic Antibody Responses in Allergic Diseases.Eur J Immunol. 2025 Mar;55(3):e202249978. doi: 10.1002/eji.202249978. Epub 2024 Dec 29. Eur J Immunol. 2025. PMID: 40071673 Free PMC article. Review.
References
-
- Flicker S, Valenta R. Renaissance of the blocking antibody concept in type I allergy. Int. Arch. Allergy Immunol. 2003;132:13–24. - PubMed
-
- Galli, S., and Lantz, C. 1999. Allergy. In Fundamental immunology. W.E. Paul, editor. Lippincott-Raven. Philadelphia, Pennsylvania, USA. 1127–1174.
-
- Sade K, Kivity S, Levy A, Fireman E. The effect of specific immunotherapy on T-cell receptor repertoire in patients with allergy to house-dust mite. Allergy. 2003;58:430–434. - PubMed
-
- van Neerven RJ, et al. Blocking antibodies induced by specific allergy vaccination prevent the activation of CD4+ T cells by inhibiting serum-IgE-facilitated allergen presentation. J. Immunol. 1999;163:2944–2952. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

