Low-temperature glass transitions of quenched and annealed bovine serum albumin aqueous solutions
- PMID: 16500968
- PMCID: PMC1440754
- DOI: 10.1529/biophysj.105.075986
Low-temperature glass transitions of quenched and annealed bovine serum albumin aqueous solutions
Abstract
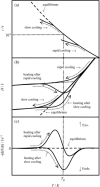
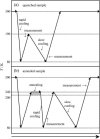
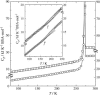
To investigate the glass transition behaviors of a 20% (w/w) aqueous solution of bovine serum albumin, heat capacities and enthalpy relaxation rates were measured by adiabatic calorimetry at temperatures ranging from 80 to 300 K. One series of measurements was carried out after quenching from 300 down to 80 K and another after annealing in 200-240 K. The quenched sample showed a heat capacity jump indicating a glass transition temperature T(g) = 170 K, and the annealed sample showed a smaller jump with the T(g) shifted toward the higher temperature side. The temperature dependence of the enthalpy relaxation rates for the quenched sample indicated the presence of two enthalpy relaxation effects: one at around 110 K and the other over a wide temperature range (120-190 K). The annealed sample showed three separate relaxation effects giving 1) T(g) = 110 K, 2) 135 K, and 3) temperature higher than 180 K, whereas nothing around 170 K. These effects were thought to originate, respectively, from the rearrangement motions of 1) primary hydrate water forming a direct hydrogen bond with the protein, 2) part of the internal water localized in the opening of a protein structure, and 3) the disordered region in the protein.
Figures


Similar articles
-
Glass transitions in aqueous solutions of protein (bovine serum albumin).J Phys Chem B. 2009 Oct 29;113(43):14448-56. doi: 10.1021/jp905511w. J Phys Chem B. 2009. PMID: 19799444
-
Hydrogen-bond network formation of water molecules and its effects on the glass transitions in the ethylene glycol aqueous solutions: failure of the Gordon-Taylor law in the water-rich range and absence of the T(g) = 115 K rearrangement process in bulk pure water.J Phys Condens Matter. 2010 Aug 18;22(32):325103. doi: 10.1088/0953-8984/22/32/325103. Epub 2010 Jul 7. J Phys Condens Matter. 2010. PMID: 21386485
-
1H- and 2H-NMR study of bovine serum albumin solutions.Biochim Biophys Acta. 1987 Sep 2;915(1):1-18. doi: 10.1016/0167-4838(87)90119-1. Biochim Biophys Acta. 1987. PMID: 3620479
-
The protein-solvent glass transition.Biochim Biophys Acta. 2010 Jan;1804(1):3-14. doi: 10.1016/j.bbapap.2009.06.019. Epub 2009 Jul 3. Biochim Biophys Acta. 2010. PMID: 19577666 Review.
-
Heat capacity and nuclear magnetic relaxation times of non-freezing water restrained by polysaccharides, revisited.J Biomater Sci Polym Ed. 2017 Jul-Aug;28(10-12):1215-1230. doi: 10.1080/09205063.2017.1291551. Epub 2017 Feb 16. J Biomater Sci Polym Ed. 2017. PMID: 28277008 Review.
Cited by
-
Analysis of Interaction Between Interfacial Structure and Fibrinogen at Blood-Compatible Polymer/Water Interface.Front Chem. 2018 Nov 8;6:542. doi: 10.3389/fchem.2018.00542. eCollection 2018. Front Chem. 2018. PMID: 30467540 Free PMC article.
-
Low-temperature polymorphic phase transition in a crystalline tripeptide L-Ala-L-Pro-Gly·H2O revealed by adiabatic calorimetry.J Phys Chem B. 2015 Feb 5;119(5):1787-92. doi: 10.1021/jp508710g. Epub 2015 Jan 27. J Phys Chem B. 2015. PMID: 25588051 Free PMC article.
-
Thermal Behavior of Water in Sephadex® G25 Gels at Low Temperatures Studied by Adiabatic Calorimetry.Gels. 2023 Feb 2;9(2):126. doi: 10.3390/gels9020126. Gels. 2023. PMID: 36826296 Free PMC article.
-
Vitrification Solutions for Plant Cryopreservation: Modification and Properties.Plants (Basel). 2021 Nov 29;10(12):2623. doi: 10.3390/plants10122623. Plants (Basel). 2021. PMID: 34961099 Free PMC article. Review.
-
Structural and dynamical examination of the low-temperature glass transition in serum albumin.Biophys J. 2006 Nov 15;91(10):3841-7. doi: 10.1529/biophysj.106.090126. Epub 2006 Aug 25. Biophys J. 2006. PMID: 16935952 Free PMC article.
References
-
- Careri, G., E. Gratton, P.-H. Yang, and J. A. Rupley. 1980. Correlation of IR spectroscopic, heat capacity, diamagnetic susceptibility and enzymatic measurements on lysozyme powder. Nature. 284:572–573. - PubMed
-
- Frauenfelder, H., S. G. Sligar, and P. G. Wolynes. 1991. The energy landscapes and motions of proteins. Science. 254:1598–1603. - PubMed
-
- Rasmussen, B. F., A. M. Stock, D. Ringe, and G. A. Petsko. 1992. Crystalline ribonuclease A loses function below the dynamical transition at 220 K. Nature. 357:423–424. - PubMed
-
- Doster, W., S. Cusack, and W. Petry. 1989. Dynamical transition of myoglobin revealed by inelastic neutron scattering. Nature. 337:754–756. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

