Preclinical evaluation of two neutralizing human monoclonal antibodies against hepatitis C virus (HCV): a potential treatment to prevent HCV reinfection in liver transplant patients
- PMID: 16501075
- PMCID: PMC1395448
- DOI: 10.1128/JVI.80.6.2654-2664.2006
Preclinical evaluation of two neutralizing human monoclonal antibodies against hepatitis C virus (HCV): a potential treatment to prevent HCV reinfection in liver transplant patients
Abstract
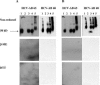
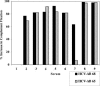
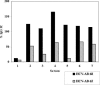
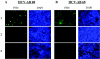
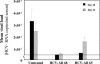
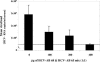
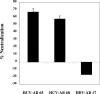
Passive immunotherapy is potentially effective in preventing reinfection of liver grafts in hepatitis C virus (HCV)-associated liver transplant patients. A combination of monoclonal antibodies directed against different epitopes may be advantageous against a highly mutating virus such as HCV. Two human monoclonal antibodies (HumAbs) against the E2 envelope protein of HCV were developed and tested for the ability to neutralize the virus and prevent human liver infection. These antibodies, designated HCV-AB 68 and HCV-AB 65, recognize different conformational epitopes on E2. They were characterized in vitro biochemically and functionally. Both HumAbs are immunoglobulin G1 and have affinity constants to recombinant E2 constructs in the range of 10(-10) M. They are able to immunoprecipitate HCV particles from infected patients' sera from diverse genotypes and to stain HCV-infected human liver tissue. Both antibodies can fix complement and form immune complexes, but they do not activate complement-dependent or antibody-dependent cytotoxicity. Upon complement fixation, the monoclonal antibodies induce phagocytosis of the immune complexes by neutrophils, suggesting that the mechanism of viral clearance includes endocytosis. In vivo, in the HCV-Trimera model, both HumAbs were capable of inhibiting HCV infection of human liver fragments and of reducing the mean viral load in HCV-positive animals. The demonstrated neutralizing activities of HCV-AB 68 and HCV-AB 65 suggest that they have the potential to prevent reinfection in liver transplant patients and to serve as prophylactic treatment in postexposure events.
Figures



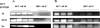
References
-
- Alter, M. J., E. E. Mast, L. A. Moyer, and H. S. Margolis. 1998. Hepatitis C. Infect. Dis. Clin. N. Am. 12:13-26. - PubMed
-
- Bartenschlager, R., and V. Lohmann. 2000. Replication of hepatitis C virus. J. Gen. Virol. 81:1631-1648. - PubMed
-
- Bartosch, B., J. Bukh, J. C. Meunier, C. Granier, R. E. Engle, W. C. Blackwelder, S. U. Emerson, F. L. Cosset, and R. H. Purcell. 2003. In vitro assay for neutralizing antibody to hepatitis C virus: evidence for broadly conserved neutralization epitopes. Proc. Natl. Acad. Sci. USA 100:14199-14204. - PMC - PubMed
-
- Bartosch, B., A. Vitelli, C. Granier, C. Goujon, J. Dubuisson, S. Pascale, E. Scarselli, R. Cortese, A. Nicosia, and F. L. Cosset. 2003. Cell entry of hepatitis C virus requires a set of co-receptors that include the CD81 tetraspanin and the SR-B1 scavenger receptor. J. Biol. Chem. 278:41624-41630. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

