Single periocular injection of celecoxib-PLGA microparticles inhibits diabetes-induced elevations in retinal PGE2, VEGF, and vascular leakage
- PMID: 16505053
- PMCID: PMC3324981
- DOI: 10.1167/iovs.05-0531
Single periocular injection of celecoxib-PLGA microparticles inhibits diabetes-induced elevations in retinal PGE2, VEGF, and vascular leakage
Abstract
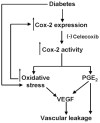
Purpose: To determine whether celecoxib inhibits VEGF secretion from ARPE-19 cells and to investigate further the safety and effectiveness of periocular celecoxib-poly (lactide-co-glycolide; PLGA) microparticles in inhibiting elevations in retinal PGE(2), VEGF, and blood-tissue barrier leakage at the end of 60 days in a streptozotocin diabetic rat model.
Methods: VEGF mRNA and protein expression in ARPE-19 cells was evaluated in the presence of 0 to 10 microM celecoxib, and cytotoxicity of celecoxib on ARPE-19 and RF6A cells was evaluated over a 0- to 100-microM concentration range. Celecoxib-PLGA microparticles were prepared by a modified solvent evaporation technique, sterilized by 25 kGy of gamma-irradiation, and characterized for size, zeta potential, drug loading, and in vitro release. Normal and streptozotocin-diabetic male Sprague-Dawley rats were divided into five groups: normal, diabetic, diabetic+placebo, normal+celecoxib, and diabetic+celecoxib. Phosphate-buffered saline (PBS) containing celecoxib-PLGA microparticles, placebo PLGA microparticles, or plain PBS in one eye was injected into the posterior subconjunctival (periocular) space in rats under anesthesia. Sixty days after administration, the animals were killed, and retinal PGE2 secretion, VEGF protein, and blood-retinal barrier leakage were estimated. Blood cell counts, blood chemistry and histology were used to assess the safety of the microparticulate system.
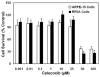
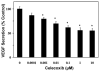
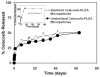
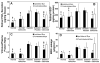
Results: Celecoxib (up to 25 microM) did not cause significant cytotoxicity in ARPE-19 or RF6A cells. Nanomolar concentrations of celecoxib reduced VEGF mRNA and VEGF protein secretion. Celecoxib-PLGA microparticles (diameter: 1140 +/- 15 nm), containing 14.93% +/- 0.21% of celecoxib sustained in vitro drug release and in vivo drug levels in the retina for 60 days. Diabetes elevated PGE2 secretion, VEGF protein, the vitreous-plasma protein ratio, and blood-retinal barrier leakage by 3-, 1.7-, 3.1-, and 2.7-fold, and celecoxib-PLGA microparticles significantly reduced these elevations by 40%, 50%, 40%, and 50%, respectively. Neither the placebo-treated eyes nor the contralateral eyes in celecoxib-PLGA microparticle-treated rats showed significant effects. Celecoxib-PLGA or placebo-PLGA particles had no effect on the body weight or blood sugar level of rats. The celecoxib-PLGA microparticles did not cause any changes in blood cell counts or chemistry and caused no histopathological damage to the retina or periocular tissues.
Conclusions: Nanomolar concentrations of celecoxib can inhibit VEGF mRNA and protein expression from ARPE-19 cells. Periocular celecoxib microparticles are useful sustained drug delivery systems for inhibiting diabetes-induced elevations in PGE2, VEGF, and blood-retinal barrier leakage. The periocular celecoxib-PLGA microparticles are safe and do not cause any damage to the retina.
Figures
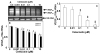


Similar articles
-
Subconjunctivally administered celecoxib-PLGA microparticles sustain retinal drug levels and alleviate diabetes-induced oxidative stress in a rat model.Eur J Pharmacol. 2005 Mar 28;511(2-3):191-8. doi: 10.1016/j.ejphar.2005.02.019. Eur J Pharmacol. 2005. PMID: 15792788
-
Celecoxib, a selective cyclooxygenase-2 inhibitor, inhibits retinal vascular endothelial growth factor expression and vascular leakage in a streptozotocin-induced diabetic rat model.Eur J Pharmacol. 2003 Jan 5;458(3):283-9. doi: 10.1016/s0014-2999(02)02793-0. Eur J Pharmacol. 2003. PMID: 12504784
-
Effect of eye pigmentation on transscleral drug delivery.Invest Ophthalmol Vis Sci. 2008 Jan;49(1):333-41. doi: 10.1167/iovs.07-0214. Invest Ophthalmol Vis Sci. 2008. PMID: 18172110 Free PMC article.
-
Subconjunctival nano- and microparticles sustain retinal delivery of budesonide, a corticosteroid capable of inhibiting VEGF expression.Invest Ophthalmol Vis Sci. 2003 Mar;44(3):1192-201. doi: 10.1167/iovs.02-0791. Invest Ophthalmol Vis Sci. 2003. PMID: 12601049
-
Delivery of celecoxib for treating diseases of the eye: influence of pigment and diabetes.Expert Opin Drug Deliv. 2010 May;7(5):631-45. doi: 10.1517/17425241003663236. Expert Opin Drug Deliv. 2010. PMID: 20205602 Free PMC article. Review.
Cited by
-
Comparison of suprachoroidal drug delivery with subconjunctival and intravitreal routes using noninvasive fluorophotometry.PLoS One. 2012;7(10):e48188. doi: 10.1371/journal.pone.0048188. Epub 2012 Oct 31. PLoS One. 2012. PMID: 23118950 Free PMC article.
-
Breakdown of the blood-ocular barrier as a strategy for the systemic use of nanosystems.Pharmaceutics. 2012 May 14;4(2):252-75. doi: 10.3390/pharmaceutics4020252. Pharmaceutics. 2012. PMID: 24300231 Free PMC article.
-
Immunological mechanisms in the pathogenesis of diabetic retinopathy.Semin Immunopathol. 2008 Apr;30(2):65-84. doi: 10.1007/s00281-008-0111-x. Epub 2008 Mar 14. Semin Immunopathol. 2008. PMID: 18340447 Review.
-
Elucidating glial responses to products of diabetes-associated systemic dyshomeostasis.Prog Retin Eye Res. 2023 May;94:101151. doi: 10.1016/j.preteyeres.2022.101151. Epub 2023 Apr 5. Prog Retin Eye Res. 2023. PMID: 37028118 Free PMC article. Review.
-
Novel Use of PLGA Microspheres to Create an Animal Model of Glaucoma with Progressive Neuroretinal Degeneration.Pharmaceutics. 2021 Feb 8;13(2):237. doi: 10.3390/pharmaceutics13020237. Pharmaceutics. 2021. PMID: 33567776 Free PMC article.
References
-
- Niitsu Y, Takayama T, Miyanishi K, et al. Chemoprevention of colorectal cancer. Cancer Chemother Pharmacol. 2004;54(suppl 1):S40–S43. - PubMed
-
- Koehne CH, Dubois RN. COX-2 inhibition and colorectal cancer. Semin Oncol. 2004;31:12–21. - PubMed
-
- Guastalla JP, Bachelot T, Ray-Coquard I. Cyclooxygenase 2 and breast cancer. From biological concepts to therapeutic trials [in French] Bull Cancer. 2004;91:S99–S108. - PubMed
-
- Spano JP, Chouahnia K, Morere JF. Cyclooxygenase 2 inhibitors and lung carcinoma [in French] Bull Cancer. 2004;91:S109–S12. - PubMed
-
- Basler JW, Piazza GA. Nonsteroidal anti-inflammatory drugs and cyclooxygenase-2 selective inhibitors for prostate cancer chemo-prevention. J Urol. 2004;171:S59–S62. discussion S62–S63. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

