Extender unit and acyl carrier protein specificity of ketosynthase domains of the 6-deoxyerythronolide B synthase
- PMID: 16506788
- PMCID: PMC2532788
- DOI: 10.1021/ja058093d
Extender unit and acyl carrier protein specificity of ketosynthase domains of the 6-deoxyerythronolide B synthase
Abstract
Polyketide synthases (PKSs) catalyze the production of numerous biologically important natural products via repeated decarboxylative condensation reactions. Modular PKSs, such as the 6-deoxyerythronolide B synthase (DEBS), consist of multiple catalytic modules, each containing a unique set of covalently linked catalytic domains. To better understand the engineering opportunities of these assembly lines, the extender unit and acyl carrier protein (ACP) specificity of keto synthase (KS) domains from modules 3 and 6 of DEBS were analyzed. These studies were undertaken with a newly developed didomain [KS][AT] construct, which lacks its own ACP domain and can therefore be interrogated with homologous or heterologous ACP or acyl-ACP substrates. By substituting the natural methylmalonyl extender unit with a malonyl group, a modest role was demonstrated for the KS in recognition of the nucleophilic substrate. The KS domain from module 3 of DEBS was found to exhibit a distinct ACP-recognition profile from the KS domain of module 6. On the basis of the above kinetic insights, a hybrid module was constructed ([KS3][AT3][KR5][ACP5][TE]) which displayed substrate recognition and elongation capabilities consistent with the natural module 3 protein. Unlike module 3, however, which lacks a ketoreductase (KR) domain, the hybrid module was able to catalyze reduction of the beta-ketothioester product of chain elongation. The high expression level and functionality of this hybrid protein demonstrates the usefulness of kinetic analysis for hybrid module design.
Figures




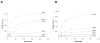

Similar articles
-
Reconstituting modular activity from separated domains of 6-deoxyerythronolide B synthase.Biochemistry. 2004 Nov 9;43(44):13892-8. doi: 10.1021/bi048418n. Biochemistry. 2004. PMID: 15518537
-
Mechanistic analysis of acyl transferase domain exchange in polyketide synthase modules.J Am Chem Soc. 2003 May 7;125(18):5366-74. doi: 10.1021/ja029539i. J Am Chem Soc. 2003. PMID: 12720450
-
Structure-based dissociation of a type I polyketide synthase module.Chem Biol. 2007 Jul;14(7):784-92. doi: 10.1016/j.chembiol.2007.05.015. Chem Biol. 2007. PMID: 17656315 Free PMC article.
-
Recognition of acyl carrier proteins by ketoreductases in assembly line polyketide synthases.J Antibiot (Tokyo). 2016 Jul;69(7):507-10. doi: 10.1038/ja.2016.41. Epub 2016 Apr 27. J Antibiot (Tokyo). 2016. PMID: 27118242 Free PMC article.
-
The structural role of the carrier protein--active controller or passive carrier.Nat Prod Rep. 2012 Oct;29(10):1111-37. doi: 10.1039/c2np20062g. Epub 2012 Aug 24. Nat Prod Rep. 2012. PMID: 22930263 Review.
Cited by
-
Discovery and Characterization of Antibody Probes of Module 2 of the 6-Deoxyerythronolide B Synthase.Biochemistry. 2023 Jun 6;62(11):1589-1593. doi: 10.1021/acs.biochem.3c00156. Epub 2023 May 15. Biochemistry. 2023. PMID: 37184546 Free PMC article.
-
Stereospecificity of ketoreductase domains 1 and 2 of the tylactone modular polyketide synthase.J Am Chem Soc. 2008 Sep 3;130(35):11598-9. doi: 10.1021/ja804453p. Epub 2008 Aug 12. J Am Chem Soc. 2008. PMID: 18693734 Free PMC article.
-
Crystal structure of the acyltransferase domain of the iterative polyketide synthase in enediyne biosynthesis.J Biol Chem. 2012 Jun 29;287(27):23203-15. doi: 10.1074/jbc.M112.362210. Epub 2012 May 15. J Biol Chem. 2012. PMID: 22589546 Free PMC article.
-
Synthetic biology of polyketide synthases.J Ind Microbiol Biotechnol. 2018 Jul;45(7):621-633. doi: 10.1007/s10295-018-2021-9. Epub 2018 Feb 9. J Ind Microbiol Biotechnol. 2018. PMID: 29423743
-
Streptomyces: host for refactoring of diverse bioactive secondary metabolites.3 Biotech. 2021 Jul;11(7):340. doi: 10.1007/s13205-021-02872-y. Epub 2021 Jun 16. 3 Biotech. 2021. PMID: 34221811 Free PMC article. Review.
References
-
- Cane DE, Walsh CT, Khosla C. Science. 1998;282:63–68. - PubMed
-
- Khosla C, Gokhale RS, Jacobsen JR, Cane DE. Annu Rev Biochem. 1999;68:219–253. - PubMed
-
- Staunton J, Weissman KJ. Nat Prod Rep. 2001;18:380–416. - PubMed
-
- Walsh CT. ChemBioChem. 2002;3:124–134.
-
- Walsh CT. Science. 2004;303:1805–1810. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

