Human scalp hair follicles are both a target and a source of prolactin, which serves as an autocrine and/or paracrine promoter of apoptosis-driven hair follicle regression
- PMID: 16507890
- PMCID: PMC1606541
- DOI: 10.2353/ajpath.2006.050468
Human scalp hair follicles are both a target and a source of prolactin, which serves as an autocrine and/or paracrine promoter of apoptosis-driven hair follicle regression
Abstract
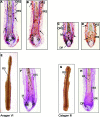
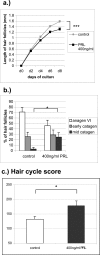
The prototypic pituitary hormone prolactin (PRL) exerts a wide variety of bioregulatory effects in mammals and is also found in extrapituitary sites, including murine skin. Here, we show by reverse transcriptase-polymerase chain reaction and immunohistology that, contrary to a previous report, human skin and normal human scalp hair follicles (HFs), in particular, express both PRL and PRL receptors (PRL-R) at the mRNA and protein level. PRL and PRL-R immunoreactivity can be detected in the epithelium of human anagen VI HFs, while the HF mesenchyme is negative. During the HF transformation from growth (anagen) to apoptosis-driven regression (catagen), PRL and PRL-R immunoreactivity appear up-regulated. Treatment of organ-cultured human scalp HFs with high-dose PRL (400 ng/ml) results in a significant inhibition of hair shaft elongation and premature catagen development, along with reduced proliferation and increased apoptosis of hair bulb keratinocytes (Ki-67/terminal dUTP nick-end labeling immunohistomorphometry). This shows that PRL receptors, expressed in HFs, are functional and that human skin and human scalp HFs are both direct targets and sources of PRL. Our data suggest that PRL acts as an autocrine hair growth modulator with catagen-promoting functions and that the hair growth-inhibitory effects of PRL demonstrated here may underlie the as yet ill-understood hair loss in patients with hyper-prolactinemia.
Figures


Similar articles
-
Thyrotropin-releasing hormone and oestrogen differentially regulate prolactin and prolactin receptor expression in female human skin and hair follicles in vitro.Br J Dermatol. 2010 May;162(5):1127-31. doi: 10.1111/j.1365-2133.2010.09676.x. Epub 2010 Mar 10. Br J Dermatol. 2010. PMID: 20302576
-
Interferon-gamma is a potent inducer of catagen-like changes in cultured human anagen hair follicles.Br J Dermatol. 2005 Apr;152(4):623-31. doi: 10.1111/j.1365-2133.2005.06453.x. Br J Dermatol. 2005. PMID: 15840090
-
Prolactin and its receptor are expressed in murine hair follicle epithelium, show hair cycle-dependent expression, and induce catagen.Am J Pathol. 2003 May;162(5):1611-21. doi: 10.1016/S0002-9440(10)64295-2. Am J Pathol. 2003. PMID: 12707045 Free PMC article.
-
Prolactin: an emerging force along the cutaneous-endocrine axis.Trends Endocrinol Metab. 2010 Sep;21(9):569-77. doi: 10.1016/j.tem.2010.06.001. Epub 2010 Jul 2. Trends Endocrinol Metab. 2010. PMID: 20598901 Review.
-
Neuroendocrinology of the hair follicle: principles and clinical perspectives.Trends Mol Med. 2014 Oct;20(10):559-70. doi: 10.1016/j.molmed.2014.06.002. Epub 2014 Jul 21. Trends Mol Med. 2014. PMID: 25066729 Review.
Cited by
-
Hormonal Effects on Hair Follicles.Int J Mol Sci. 2020 Jul 28;21(15):5342. doi: 10.3390/ijms21155342. Int J Mol Sci. 2020. PMID: 32731328 Free PMC article. Review.
-
Hair Loss in a Hemodialysis Patient after Repetitive Use of the Antipruritic Drug Nalfurafine: Implications of Impaired Angiogenesis for Hair Loss.Intern Med. 2025 May 1;64(9):1371-1379. doi: 10.2169/internalmedicine.4249-24. Epub 2024 Oct 11. Intern Med. 2025. PMID: 39401915 Free PMC article.
-
Chitosan Nanoparticles for Topical Drug Delivery in Chemotherapy-Induced Alopecia: A Comparative Study of Five Repurposed Pharmacological Agents.Pharmaceuticals (Basel). 2025 Jul 21;18(7):1071. doi: 10.3390/ph18071071. Pharmaceuticals (Basel). 2025. PMID: 40732359 Free PMC article.
-
The quantification of reproductive hormones in the hair of captive adult brown bears and their application as indicators of sex and reproductive state.Conserv Physiol. 2017 Jun 1;5(1):cox032. doi: 10.1093/conphys/cox032. eCollection 2017. Conserv Physiol. 2017. PMID: 28580147 Free PMC article.
-
Completely humanizing prolactin rescues infertility in prolactin knockout mice and leads to human prolactin expression in extrapituitary mouse tissues.Endocrinology. 2013 Dec;154(12):4777-89. doi: 10.1210/en.2013-1476. Epub 2013 Sep 12. Endocrinology. 2013. PMID: 24029242 Free PMC article.
References
-
- Freeman M, Kanycisca B, Lerant A, Nagy G. Prolactin: structure, function and regulation of secretion. Physiol Rev. 2000;80:1523–1631. - PubMed
-
- Goldhar AS, Vonderhaar BK, Trott JF, Hovey RC. Prolactin-induced expression of vascular endothelial growth factor via Egr-1. Mol Cell Endocrinol. 2005;232:9–19. - PubMed
-
- Gootwine E. Placental hormones and fetal-placental development. Anim Reprod Sci. 2004;82–83:551–566. - PubMed
-
- Larsen PR, Kronenberg HM, Melmed S, Polonsky KS, editors. Philadelphia: Saunders; Williams Textbook of Endocrinology. (ed 10.) 2003
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

