Inhibition of interferon-gamma signaling in oligodendroglia delays coronavirus clearance without altering demyelination
- PMID: 16507895
- PMCID: PMC1606538
- DOI: 10.2353/ajpath.2006.050496
Inhibition of interferon-gamma signaling in oligodendroglia delays coronavirus clearance without altering demyelination
Abstract
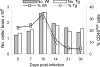
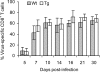
Infection of the central nervous system (CNS) by the neurotropic JHM strain of mouse hepatitis virus (JHMV) induces an acute encephalomyelitis associated with demyelination. To examine the anti-viral and/or regulatory role of interferon-gamma (IFN-gamma) signaling in the cell that synthesizes and maintains the myelin sheath, we analyzed JHMV pathogenesis in transgenic mice expressing a dominant-negative IFN-gamma receptor on oligodendroglia. Defective IFN-gamma signaling was associated with enhanced oligodendroglial tropism and delayed virus clearance. However, the CNS inflammatory cell composition and CD8(+) T-cell effector functions were similar between transgenic and wild-type mice, supporting unimpaired peripheral and CNS immune responses in transgenic mice. Surprisingly, increased viral load in oligodendroglia did not affect the extent of myelin loss, the frequency of oligodendroglial apoptosis, or CNS recruitment of macrophages. These data demonstrate that IFN-gamma receptor signaling is critical for the control of JHMV replication in oligodendroglia. In addition, the absence of a correlation between increased oligodendroglial infection and the extent of demyelination suggests a complex pathobiology of myelin loss in which infection of oligodendroglia is required but not sufficient.
Figures

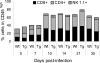


Similar articles
-
Perforin and gamma interferon-mediated control of coronavirus central nervous system infection by CD8 T cells in the absence of CD4 T cells.J Virol. 2004 Feb;78(4):1739-50. doi: 10.1128/jvi.78.4.1739-1750.2004. J Virol. 2004. PMID: 14747539 Free PMC article.
-
IFN-gamma is required for viral clearance from central nervous system oligodendroglia.J Immunol. 1999 Feb 1;162(3):1641-7. J Immunol. 1999. PMID: 9973424
-
Gamma interferon signaling in oligodendrocytes is critical for protection from neurotropic coronavirus infection.J Virol. 2010 Mar;84(6):3111-5. doi: 10.1128/JVI.02373-09. Epub 2009 Dec 30. J Virol. 2010. PMID: 20042510 Free PMC article.
-
Chemokine CXCL10 and Coronavirus-Induced Neurologic Disease.Viral Immunol. 2019 Jan/Feb;32(1):25-37. doi: 10.1089/vim.2018.0073. Epub 2018 Aug 15. Viral Immunol. 2019. PMID: 30109979 Free PMC article. Review.
-
Pathogenesis of acute and chronic central nervous system infection with variants of mouse hepatitis virus, strain JHM.Immunol Res. 2007;39(1-3):160-72. doi: 10.1007/s12026-007-0079-y. Immunol Res. 2007. PMID: 17917063 Free PMC article. Review.
Cited by
-
Neuroimmunology of central nervous system viral infections: the cells, molecules and mechanisms involved.Curr Opin Pharmacol. 2008 Aug;8(4):472-9. doi: 10.1016/j.coph.2008.05.002. Epub 2008 Jun 16. Curr Opin Pharmacol. 2008. PMID: 18562249 Free PMC article. Review.
-
Ependymal cells: roles in central nervous system infections and therapeutic application.J Neuroinflammation. 2024 Oct 9;21(1):255. doi: 10.1186/s12974-024-03240-2. J Neuroinflammation. 2024. PMID: 39385253 Free PMC article. Review.
-
Oligodendroglia are limited in type I interferon induction and responsiveness in vivo.Glia. 2012 Oct;60(10):1555-66. doi: 10.1002/glia.22375. Epub 2012 Jun 26. Glia. 2012. PMID: 22736486 Free PMC article.
-
Immune responses to non-tumor antigens in the central nervous system.Front Oncol. 2014 Nov 13;4:328. doi: 10.3389/fonc.2014.00328. eCollection 2014. Front Oncol. 2014. PMID: 25431758 Free PMC article. Review.
-
Enhanced antiviral T cell function in the absence of B7-H1 is insufficient to prevent persistence but exacerbates axonal bystander damage during viral encephalomyelitis.J Immunol. 2010 Nov 1;185(9):5607-18. doi: 10.4049/jimmunol.1001984. Epub 2010 Sep 27. J Immunol. 2010. PMID: 20876353 Free PMC article.
References
-
- Tyler KL, Gonzalez-Scarano F. Viral diseases of the central nervous system. Nathanson N, editor. Philadelphia: Lippincott-Raven; 1996:pp 837–855.
-
- Doherty PC, Allan JE, Lynch F, Ceredig R. Dissection of an inflammatory process induced by CD8+ T cells. Immunol Today. 1990;11:55–59. - PubMed
-
- Kagi D, Vignaux F, Ledermann B, Burki K, Depraetere V, Nagata S, Hengartner H, Golstein P. Fas and perforin pathways as major mechanisms of T cell-mediated cytotoxicity. Science. 1994;265:528–530. - PubMed
-
- Oldstone MB, Blount P, Southern PJ, Lampert PW. Cytoimmunotherapy for persistent virus infection reveals a unique clearance pattern from the central nervous system. Nature. 1986;321:239–243. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

