Distinct compartmentalization of CD4+ T-cell effector function versus proliferative capacity during pulmonary cryptococcosis
- PMID: 16507900
- PMCID: PMC1606518
- DOI: 10.2353/ajpath.2006.050522
Distinct compartmentalization of CD4+ T-cell effector function versus proliferative capacity during pulmonary cryptococcosis
Abstract
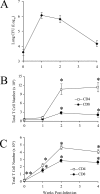
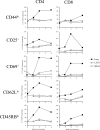
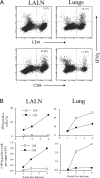
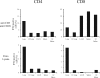
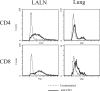
The activation and expansion of T cells and their acquisition of effector function are key steps in the development of the adaptive immune response. Most infections are predominantly outside of the lymphoid tissues, and it is unclear at what point developmentally and anatomically T cells acquire effector function in vivo. In these studies, we compared the activation and polarization of T cells during murine pulmonary Cryptococcus neoformans infection in the secondary lymphoid tissues and at the site of primary infection. Few CD4(+) and CD8(+) T cells expressed an activated phenotype (CD44(hi,) CD25(+), CD69(+), CD62L(lo), CD45RB(lo)) at the sites of clonal expansion (lymph nodes, spleen, and blood). In contrast, a high percentage of T cells expressed activation markers at the site of primary infection, the lungs. Additionally, the polarization of CD4(+) T cells to interferon-gamma-producing effector cells occurred at the site of infection, the lungs. CD4(+) and CD8(+) T cells from secondary lymphoid organs responded to TCR restimulation by proliferating, whereas T cells from the lungs proliferated poorly. This report demonstrates for the first time that T-cell activation and effector function in secondary lymphoid tissues during fungal infection is characteristically different from that at the site of primary infection.
Figures

Similar articles
-
Diversity of the T-cell response to pulmonary Cryptococcus neoformans infection.Infect Immun. 2006 Aug;74(8):4538-48. doi: 10.1128/IAI.00080-06. Infect Immun. 2006. PMID: 16861640 Free PMC article.
-
Generation of antifungal effector CD8+ T cells in the absence of CD4+ T cells during Cryptococcus neoformans infection.J Immunol. 2005 Jun 15;174(12):7920-8. doi: 10.4049/jimmunol.174.12.7920. J Immunol. 2005. PMID: 15944298
-
Immunologic homeostasis during infection: coexistence of strong pulmonary cell-mediated immunity to secondary Cryptococcus neoformans infection while the primary infection still persists at low levels in the lungs.J Immunol. 2006 Oct 1;177(7):4652-61. doi: 10.4049/jimmunol.177.7.4652. J Immunol. 2006. PMID: 16982904
-
Protective immunity against experimental pulmonary cryptococcosis in T cell-depleted mice.Clin Vaccine Immunol. 2011 May;18(5):717-23. doi: 10.1128/CVI.00036-11. Epub 2011 Mar 30. Clin Vaccine Immunol. 2011. PMID: 21450975 Free PMC article.
-
Effect of a CD4-depleting antibody on the development of Cryptococcus neoformans-induced allergic bronchopulmonary mycosis in mice.Infect Immun. 2006 Jul;74(7):4339-48. doi: 10.1128/IAI.01989-05. Infect Immun. 2006. PMID: 16790808 Free PMC article.
Cited by
-
State of the Field: Cytotoxic Immune Cell Responses in C. neoformans and C. deneoformans Infection.J Fungi (Basel). 2024 Oct 12;10(10):712. doi: 10.3390/jof10100712. J Fungi (Basel). 2024. PMID: 39452664 Free PMC article. Review.
-
Adaptive Immunity to Cryptococcus neoformans Infections.J Fungi (Basel). 2017;3(4):64. doi: 10.3390/jof3040064. Epub 2017 Nov 21. J Fungi (Basel). 2017. PMID: 29333430 Free PMC article.
-
In pulmonary paracoccidioidomycosis IL-10 deficiency leads to increased immunity and regressive infection without enhancing tissue pathology.PLoS Negl Trop Dis. 2013 Oct 24;7(10):e2512. doi: 10.1371/journal.pntd.0002512. eCollection 2013. PLoS Negl Trop Dis. 2013. PMID: 24205424 Free PMC article.
-
Cancer systems immunology reveals myeloid-T cell interactions and B cell activation mediate response to checkpoint inhibition in metastatic breast cancer.bioRxiv [Preprint]. 2025 Jun 13:2025.06.09.658361. doi: 10.1101/2025.06.09.658361. bioRxiv. 2025. PMID: 40661534 Free PMC article. Preprint.
-
Transcriptional profiling of a fungal granuloma reveals a low metabolic activity of Paracoccidioides brasiliensis yeasts and an actively regulated host immune response.Front Cell Infect Microbiol. 2023 Oct 5;13:1268959. doi: 10.3389/fcimb.2023.1268959. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 37868350 Free PMC article.
References
-
- van Stipdonk MJ, Lemmens EE, Schoenberger SP. Naive CTLs require a single brief period of antigenic stimulation for clonal expansion and differentiation. Nat Immunol. 2001;2:423–429. - PubMed
-
- Feldmesser M, Tucker S, Casadevall A. Intracellular parasitism of macrophages by Cryptococcus neoformans. Trends Microbiol. 2001;9:273–278. - PubMed
-
- Huffnagle GB, Lipscomb MF, Lovchik JA, Hoag KA, Street NE. The role of CD4+ and CD8+ T cells in the protective inflammatory response to a pulmonary cryptococcal infection. J Leukoc Biol. 1994;55:35–42. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

