VEGF is upregulated by hypoxia-induced mitogenic factor via the PI-3K/Akt-NF-kappaB signaling pathway
- PMID: 16512910
- PMCID: PMC1434739
- DOI: 10.1186/1465-9921-7-37
VEGF is upregulated by hypoxia-induced mitogenic factor via the PI-3K/Akt-NF-kappaB signaling pathway
Abstract
Background: Hypoxia-induced mitogenic factor (HIMF) is developmentally regulated and plays an important role in lung pathogenesis. We initially found that HIMF promotes vascular tubule formation in a matrigel plug model. In this study, we investigated the mechanisms which HIMF enhances expression of vascular endothelial growth factor (VEGF) in lung tissues and epithelial cells.
Methods: Recombinant HIMF protein was intratracheally instilled into adult mouse lungs, VEGF expression was examined by immunohistochemical staining and Western blot. The promoter-luciferase reporter assay, RT-PCR, and Western blot were performed to examine the effects of HIMF on VEGF expression in mouse lung epithelial cell line MLE-12. The activation of NF-kappa B (NF-kappaB) and phosphorylation of Akt, IKK and IkappaBalpha were examined by luciferase assay and Western blot, respectively.
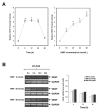
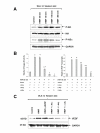
Results: Intratracheal instillation of HIMF protein resulted in significant increase of VEGF, mainly localized to airway epithelial and alveolar type II cells. Deletion of NF-kappaB binding sites within VEGF promoter abolished HIMF-induced VEGF expression in MLE-12 cells, suggesting that activation of NF-kappaB is essential for VEGF upregulation induced by HIMF. Stimulation of lung epithelial cells by HIMF resulted in phosphorylation of IKK and IkappaBalpha, leading to activation of NF-kappaB. In addition, HIMF strongly induced Akt phosphorylation, and suppression of Akt activation by specific inhibitors and dominant negative mutants for PI-3K, and IKK or IkappaBalpha blocked HIMF-induced NF-kappaB activation and attenuated HIMF-induced VEGF production.
Conclusion: These results suggest that HIMF enhances VEGF production in mouse lung epithelial cells in a PI-3K/Akt-NF-kappaB signaling pathway-dependent manner, and may play critical roles in pulmonary angiogenesis.
Figures






Similar articles
-
Participation of the PI-3K/Akt-NF-kappa B signaling pathways in hypoxia-induced mitogenic factor-stimulated Flk-1 expression in endothelial cells.Respir Res. 2006 Jul 27;7(1):101. doi: 10.1186/1465-9921-7-101. Respir Res. 2006. PMID: 16872509 Free PMC article.
-
Hypoxia-induced mitogenic factor enhances angiogenesis by promoting proliferation and migration of endothelial cells.Exp Cell Res. 2006 Nov 1;312(18):3559-69. doi: 10.1016/j.yexcr.2006.07.024. Epub 2006 Aug 9. Exp Cell Res. 2006. PMID: 16982054
-
Hypoxia-induced mitogenic factor promotes vascular adhesion molecule-1 expression via the PI-3K/Akt-NF-kappaB signaling pathway.Am J Respir Cell Mol Biol. 2006 Oct;35(4):444-56. doi: 10.1165/rcmb.2005-0424OC. Epub 2006 May 18. Am J Respir Cell Mol Biol. 2006. PMID: 16709959 Free PMC article.
-
Regulation and function of IKK and IKK-related kinases.Sci STKE. 2006 Oct 17;2006(357):re13. doi: 10.1126/stke.3572006re13. Sci STKE. 2006. PMID: 17047224 Review.
-
Insulin/IGF-1 paradox of aging: regulation via AKT/IKK/NF-kappaB signaling.Cell Signal. 2010 Apr;22(4):573-7. doi: 10.1016/j.cellsig.2009.10.006. Epub 2009 Oct 25. Cell Signal. 2010. PMID: 19861158 Review.
Cited by
-
Zinc-deficient diet aggravates ventilation-induced lung injury in rats.J Biomed Res. 2012 Jan;26(1):59-65. doi: 10.1016/S1674-8301(12)60008-6. J Biomed Res. 2012. PMID: 23554731 Free PMC article.
-
RLN2 Is a Positive Regulator of AKT-2-Induced Gene Expression Required for Osteosarcoma Cells Invasion and Chemoresistance.Biomed Res Int. 2015;2015:147468. doi: 10.1155/2015/147468. Epub 2015 Jul 1. Biomed Res Int. 2015. PMID: 26229955 Free PMC article.
-
PI3K-γ inhibition ameliorates acute lung injury through regulation of IκBα/NF-κB pathway and innate immune responses.J Clin Immunol. 2012 Apr;32(2):340-51. doi: 10.1007/s10875-011-9628-1. Epub 2011 Dec 24. J Clin Immunol. 2012. PMID: 22198681
-
Nuclear factor-kappa-B signaling in lung development and disease: one pathway, numerous functions.Birth Defects Res A Clin Mol Teratol. 2014 Mar;100(3):202-16. doi: 10.1002/bdra.23233. Epub 2014 Mar 17. Birth Defects Res A Clin Mol Teratol. 2014. PMID: 24639404 Free PMC article. Review.
-
Hypoxia-induced mitogenic factor (HIMF/FIZZ1/RELMα) in chronic hypoxia- and antigen-mediated pulmonary vascular remodeling.Respir Res. 2013 Jan 4;14(1):1. doi: 10.1186/1465-9921-14-1. Respir Res. 2013. PMID: 23289668 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

