Combinations of CD45 isoforms are crucial for immune function and disease
- PMID: 16517710
- PMCID: PMC2619577
- DOI: 10.4049/jimmunol.176.6.3417
Combinations of CD45 isoforms are crucial for immune function and disease
Abstract
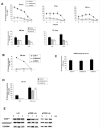
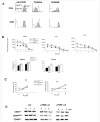
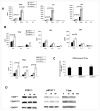
Expression of the CD45 Ag in hemopoietic cells is essential for normal development and function of lymphocytes, and both mice and humans lacking expression exhibit SCID. Human genetic variants of CD45, the exon 4 C77G and exon 6 A138G alleles, which alter the pattern of CD45 isoform expression, are associated with autoimmune and infectious diseases. We constructed transgenic mice expressing either an altered level or combination of CD45 isoforms. We show that the total level of CD45 expressed is crucial for normal TCR signaling, lymphocyte proliferation, and cytokine production. Most importantly, transgenic lines with a normal level, but altered combinations of CD45 isoforms, CD45(RABC/+) and CD45(RO/+) mice, which mimic variant CD45 expression in C77G and A138G humans, show more rapid onset and increased severity of experimental autoimmune encephalomyelitis. CD45(RO/+) cells produce more TNF-alpha and IFN-gamma. Thus, for the first time, we have shown experimentally that it is the combination of CD45 isoforms that affects immune function and disease.
Figures



Similar articles
-
Disease associations and altered immune function in CD45 138G variant carriers.Hum Mol Genet. 2004 Oct 15;13(20):2377-84. doi: 10.1093/hmg/ddh276. Epub 2004 Aug 27. Hum Mol Genet. 2004. PMID: 15333587
-
Altered CD45 isoform expression in C77G carriers influences cytokine responsiveness and adhesion properties of T cells.Clin Exp Immunol. 2007 Dec;150(3):509-17. doi: 10.1111/j.1365-2249.2007.03508.x. Epub 2007 Sep 28. Clin Exp Immunol. 2007. PMID: 17903220 Free PMC article.
-
Resident and infiltrating central nervous system APCs regulate the emergence and resolution of experimental autoimmune encephalomyelitis.J Immunol. 2001 Apr 15;166(8):5168-75. doi: 10.4049/jimmunol.166.8.5168. J Immunol. 2001. PMID: 11290800
-
A study on CD45 isoform expression during T-cell development and selection events in the human thymus.Hum Immunol. 2002 May;63(5):394-404. doi: 10.1016/s0198-8859(02)00379-8. Hum Immunol. 2002. PMID: 11975983
-
Expression of the long form of human FLIP by retroviral gene transfer of hemopoietic stem cells exacerbates experimental autoimmune encephalomyelitis.J Immunol. 2003 Feb 15;170(4):2064-73. doi: 10.4049/jimmunol.170.4.2064. J Immunol. 2003. PMID: 12574377
Cited by
-
IDEIS: a tool to identify PTPRC/CD45 isoforms from single-cell transcriptomic data.Front Immunol. 2024 Oct 9;15:1446931. doi: 10.3389/fimmu.2024.1446931. eCollection 2024. Front Immunol. 2024. PMID: 39445006 Free PMC article.
-
Get Spliced: Uniting Alternative Splicing and Arthritis.Int J Mol Sci. 2024 Jul 25;25(15):8123. doi: 10.3390/ijms25158123. Int J Mol Sci. 2024. PMID: 39125692 Free PMC article. Review.
-
The influence of three-dimensional structure on naïve T cell homeostasis and aging.Front Aging. 2022 Nov 7;3:1045648. doi: 10.3389/fragi.2022.1045648. eCollection 2022. Front Aging. 2022. PMID: 36419548 Free PMC article.
-
Unusual case presentations associated with the CD45 C77G polymorphism.Clin Exp Immunol. 2006 Dec;146(3):448-54. doi: 10.1111/j.1365-2249.2006.03230.x. Clin Exp Immunol. 2006. PMID: 17100764 Free PMC article.
-
Leucocyte common antigen (CD45) and CD5 positivity in an "undifferentiated" carcinoma: a potential diagnostic pitfall.J Clin Pathol. 2007 Aug;60(8):936-8. doi: 10.1136/jcp.2006.044750. J Clin Pathol. 2007. PMID: 17660336 Free PMC article. No abstract available.
References
-
- Hermiston ML, Xu Z, Weiss A. CD45: a critical regulator of signaling thresholds in immune cells. Annu. Rev. Immunol. 2003;21:107–137. - PubMed
-
- Penninger JM, Irie-Sasaki J, Sasaki T, Oliveira-Dos-Santos AJ. CD45: new jobs for an old acquaintance. Nat. Immunol. 2001;2:389–396. - PubMed
-
- Akbar AN, Terry L, Timms A, Beverley PC, Janossy G. Loss of CD45R and gain of UCHL1 reactivity is a feature of primed T cells. J. Immunol. 1988;140:2171–2178. - PubMed
-
- Powrie F, Mason D. Subsets of rat CD4+ T cells defined by their differential expression of variants of the CD45 antigen: developmental relationships and in vitro and in vivo functions. Curr. Top. Microbiol. Immunol. 1990;159:79–96. - PubMed
-
- Merkenschlager M, Terry L, Edwards R, Beverley PC. Limiting dilution analysis of proliferative responses in human lymphocyte populations defined by the monoclonal antibody UCHL1: implications for differential CD45 expression in T cell memory formation. Eur. J. Immunol. 1988;18:1653–1661. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

