Analysis of the antibody response to an immunodominant epitope of the envelope glycoprotein of a lentivirus and its diagnostic potential
- PMID: 16517887
- PMCID: PMC1393135
- DOI: 10.1128/JCM.44.3.981-991.2006
Analysis of the antibody response to an immunodominant epitope of the envelope glycoprotein of a lentivirus and its diagnostic potential
Abstract
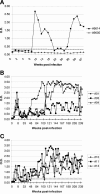
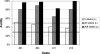
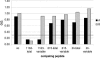
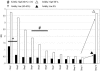
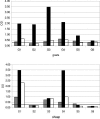
The envelope glycoprotein of small ruminant lentiviruses (SRLV) is a major target of the humoral immune response and contains several linear B-cell epitopes. We amplified and sequenced the genomic segment encoding the SU5 antigenic site of the envelope glycoprotein of several SRLV field isolates. With synthetic peptides based on the deduced amino acid sequences of SU5 in an enzyme-linked immunosorbent assay (ELISA), we have (i) proved the immunodominance of this region regardless of its high variability, (ii) defined the epitopes encompassed by SU5, (iii) illustrated the rapid and peculiar kinetics of seroconversion to this antigenic site, and (iv) shown the rapid and strong maturation of the avidity of the anti-SU5 antibody. Finally, we demonstrated the modular diagnostic potential of SU5 peptides. Under Swiss field conditions, the SU5 ELISA was shown to detect the majority of infected animals and, when applied in a molecular epidemiological context, to permit rapid phylogenetic classification of the infecting virus.
Figures

Similar articles
-
An Immunodominant Region of the Envelope Glycoprotein of Small Ruminant Lentiviruses May Function as Decoy Antigen.Viruses. 2018 May 2;10(5):231. doi: 10.3390/v10050231. Viruses. 2018. PMID: 29724026 Free PMC article.
-
Characterization of an immunodominant epitope of small ruminant lentivirus (SRLV) nucleoprotein.Vet Immunol Immunopathol. 2008 Oct 15;125(3-4):361-7. doi: 10.1016/j.vetimm.2008.05.013. Epub 2008 May 23. Vet Immunol Immunopathol. 2008. PMID: 18584880
-
Development of recombinant capsid antigen/transmembrane epitope fusion proteins for serological diagnosis of animal lentivirus infections.J Virol Methods. 2004 Oct;121(1):73-8. doi: 10.1016/j.jviromet.2004.06.001. J Virol Methods. 2004. PMID: 15350735
-
Diagnostic tests for small ruminant lentiviruses.Vet Microbiol. 2005 Apr 25;107(1-2):49-62. doi: 10.1016/j.vetmic.2005.01.012. Vet Microbiol. 2005. PMID: 15795077 Review.
-
Prevention strategies against small ruminant lentiviruses: an update.Vet J. 2009 Oct;182(1):31-7. doi: 10.1016/j.tvjl.2008.05.008. Epub 2008 Aug 27. Vet J. 2009. PMID: 18755622 Review.
Cited by
-
Public Baseline and shared response structures support the theory of antibody repertoire functional commonality.PLoS Comput Biol. 2021 Mar 1;17(3):e1008781. doi: 10.1371/journal.pcbi.1008781. eCollection 2021 Mar. PLoS Comput Biol. 2021. PMID: 33647011 Free PMC article.
-
Eradication of caprine arthritis encephalitis virus in the goat population of South Tyrol, Italy: analysis of the tailing phenomenon during the 2016-2017 campaign.J Vet Diagn Invest. 2020 Jul;32(4):589-593. doi: 10.1177/1040638720934055. Epub 2020 Jun 18. J Vet Diagn Invest. 2020. PMID: 32552528 Free PMC article.
-
The genetic variability of small-ruminant lentiviruses and its impact on tropism, the development of diagnostic tests and vaccines and the effectiveness of control programmes.J Vet Res. 2023 Dec 19;67(4):479-502. doi: 10.2478/jvetres-2023-0064. eCollection 2023 Dec. J Vet Res. 2023. PMID: 38130459 Free PMC article.
-
Serological, Molecular and Culture-Based Diagnosis of Lentiviral Infections in Small Ruminants.Viruses. 2021 Aug 27;13(9):1711. doi: 10.3390/v13091711. Viruses. 2021. PMID: 34578292 Free PMC article. Review.
-
Demonstration of coinfection with and recombination by caprine arthritis-encephalitis virus and maedi-visna virus in naturally infected goats.J Virol. 2007 May;81(10):4948-55. doi: 10.1128/JVI.00126-07. Epub 2007 Mar 7. J Virol. 2007. PMID: 17344293 Free PMC article.
References
-
- Barin, F., Y. Lahbabi, L. Buzelay, B. Lejeune, A. Baillou-Beaufils, F. Denis, C. Mathiot, S. M'Boup, V. Vithayasai, U. Dietrich, and A. Goudeau. 1996. Diversity of antibody binding to V3 peptides representing consensus sequences of HIV type 1 genotypes A to E: an approach for HIV type 1 serological subtyping. AIDS Res. Hum. Retrovir. 12:1279-1289. - PubMed
-
- Bertoni, G., C. Hertig, M. L. Zahno, H. R. Vogt, S. Dufour, P. Cordano, E. Peterhans, W. P. Cheevers, P. Sonigo, and G. Pancino. 2000. B-cell epitopes of the envelope glycoprotein of caprine arthritis-encephalitis virus and antibody response in infected goats. J. Gen. Virol. 81:2929-2940. - PubMed
-
- Bertoni, G., M.-L. Zahno, R. Zanoni, H. R. Vogt, E. Peterhans, G. Ruff, W. P. Cheevers, P. Sonigo, and G. Pancino. 1994. Antibody reactivity to the immunodominant epitopes of the caprine arthritis-encephalitis virus gp38 transmembrane protein associates with the development of arthritis. J. Virol. 68:7139-7147. - PMC - PubMed
-
- de Andres, D., D. Klein, N. J. Watt, E. Berriatua, S. Torsteinsdottir, B. A. Blacklaws, and G. D. Harkiss. 2005. Diagnostic tests for small ruminant lentiviruses. Vet. Microbiol. 107:49-62. - PubMed
-
- Fevereiro, M., S. Barros, and T. Fagulha. 1999. Development of a monoclonal antibody blocking-ELISA for detection of antibodies against maedi-Visna virus. J. Virol. Methods 81:101-108. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

