A direct interaction with calponin inhibits the actin-nucleating activity of gelsolin
- PMID: 16536729
- PMCID: PMC1482823
- DOI: 10.1042/BJ20051690
A direct interaction with calponin inhibits the actin-nucleating activity of gelsolin
Abstract
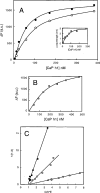
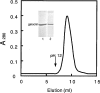
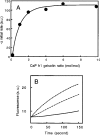
Gelsolin and calponin are well-characterized cytoskeletal proteins that are abundant and widely expressed in vertebrate tissues. It is also becoming apparent, however, that they are involved in cell signalling. In the present study, we show that gelsolin and calponin interact directly to form a high-affinity (K(d)=16 nM) 1:1 complex, by the use of fluorescent probes attached to both proteins, by affinity chromatography and by immunoprecipitation. These methods show that gelsolin can form high-affinity complexes with two calponin isoforms (basic h1 and acidic h3). They also show that gelsolin binds calponin through regions that have been identified previously as being calponin's actin-binding sites. Moreover, gelsolin does not interact with calponin while calponin is bound to F-actin. Reciprocal experiments to find calponin-binding sites on gelsolin show that these are in both the N- and C-terminal halves of gelsolin. Calponin has minimal effects on actin severing by gelsolin. In contrast, calponin markedly affects the nucleation activity of gelsolin. The maximum inhibition of nucleation by gelsolin was 50%, which was achieved with a ratio of two calponins for every gelsolin. Thus the interaction of calponin with gelsolin may play a regulatory role in the formation of actin filaments through modulation of gelsolin's actin-binding function and through the prevention of calponin's actin-binding activities.
Figures






Similar articles
-
The gelsolin:calponin complex nucleates actin filaments with distinct morphologies.Biochem Biophys Res Commun. 2010 Feb 5;392(2):118-23. doi: 10.1016/j.bbrc.2009.12.103. Epub 2009 Dec 24. Biochem Biophys Res Commun. 2010. PMID: 20035726
-
Two distinct sites of interaction form the calponin: gelsolin complex and two calcium switches control its activity.Biochim Biophys Acta. 2007 Jul;1774(7):952-8. doi: 10.1016/j.bbapap.2007.04.015. Epub 2007 May 6. Biochim Biophys Acta. 2007. PMID: 17556051
-
Calponin binds G-actin and F-actin with similar affinity.FEBS Lett. 2006 Sep 4;580(20):4801-6. doi: 10.1016/j.febslet.2006.07.065. Epub 2006 Aug 4. FEBS Lett. 2006. PMID: 16901482
-
Novel structural insights into F-actin-binding and novel functions of calponin homology domains.Curr Opin Struct Biol. 2008 Dec;18(6):702-8. doi: 10.1016/j.sbi.2008.10.003. Epub 2008 Nov 13. Curr Opin Struct Biol. 2008. PMID: 18952167 Review.
-
Calponins: adaptable modular regulators of the actin cytoskeleton.Int J Biochem Cell Biol. 2008;40(10):1990-5. doi: 10.1016/j.biocel.2007.07.010. Epub 2007 Jul 31. Int J Biochem Cell Biol. 2008. PMID: 17768079 Review.
Cited by
-
Deletion of calponin 2 in macrophages attenuates the severity of inflammatory arthritis in mice.Am J Physiol Cell Physiol. 2016 Oct 1;311(4):C673-C685. doi: 10.1152/ajpcell.00331.2015. Epub 2016 Aug 3. Am J Physiol Cell Physiol. 2016. PMID: 27488671 Free PMC article.
-
Evolution and function of calponin and transgelin.Front Cell Dev Biol. 2023 Jun 8;11:1206147. doi: 10.3389/fcell.2023.1206147. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37363722 Free PMC article. Review.
References
-
- dos Remedios C. G., Chhabra D., Kekic M., Dedova I. V., Tsubakihara M., Berry D. A., Noseworthy N. J. Actin binding proteins: regulation of cytoskeletal microfilaments. Physiol. Rev. 2003;83:433–473. - PubMed
-
- Yin H. L., Stossel T. P. Control of cytoplasmic actin gel–sol transformation by gelsolin, a calcium dependent regulatory protein. Nature (London) 1979;281:583–586. - PubMed
-
- Lamb J. A., Allen P. G., Tuan B. Y., Janmey P. A. Modulation of gelsolin function: activation at low pH overrides Ca2+ requirement. J. Biol. Chem. 1993;268:8999–9004. - PubMed
-
- Lagarrigue E., Ternent D., Maciver S. K., Fattoum A., Benyamin Y., Roustan C. The activation of gelsolin by low pH: the calcium latch is sensitive to calcium but not pH. Eur. J. Biochem. 2003;270:4105–4112. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

